Table of contents
In 2015 we published a collection of FDA GMP warning letters that included deficiencies in data governance and data integrity. Here we provide the same information for 2016 drug GMP warning letters. These should serve as a resource for GMP audit staff and QA staff as they evaluate their own firms and contract sites for gaps in these areas. In this introduction, we provide tables and graphs identifying the dates of the warning letters, firms to which they were issued, and the country where the facility is located. We also provide a table that shows the trends over time for location of countries where facilities receiving these warning letters were located, beginning in 2008. And, finally, we provide a tabulation of the regulations most frequently cited for these deficiencies in 2015 and 2016.
Table 1 lists the warning letters that include data integrity deficiencies, the date of issuance, and the country where the facility is located. Note that the first two warning letters were from inspections in 2015 but published in 2016 and were not counted in last years’ tally, so I included them here. I’ve color coded the country column and have included all European countries in a single group.
Figure 1: Data Integrity Warning Letters by Country
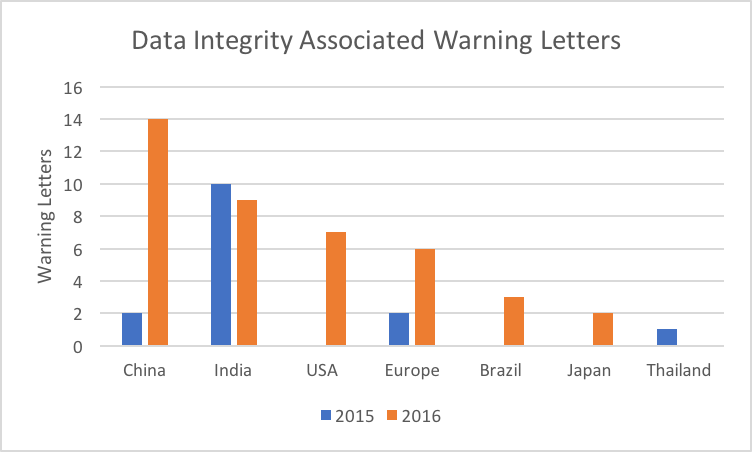
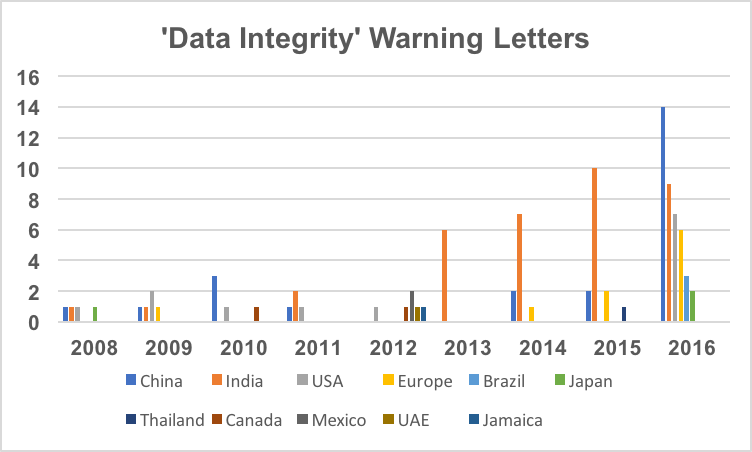
Figure 2 captures data from 2008 through 2016 on a country by country basis. Some countries are consistently present in multiple years including the US, China and India and the ‘Europe’ category. A few countries are present only in one or two years. Note that the US has warning letters with data integrity deficiencies in all years except 2013, 2014, and 2015 and is in fact the third most frequent country for 2016 warning letters of this type.
Figure 2
Conclusion:
In 2017 I look for much of the same in terms of warning letter deficiencies in the area of data integrity. I would expect the number of warning letters issued to China to continue to increase assuming foreign inspections are not curtailed as part of potential cost cutting under the new administration. I would also expect to see more focus on electronic records issues in batch records and in the manufacturing area as FDA expands their focus out from the laboratory. Compliance in this area continues to bedevil the industry, both domestic and foreign manufacturing sites. Perhaps one potential remedy is to have firms’ internal GMP audits and assessments focus in depth on this area. Remediation here often takes time and can be expensive if new instrumentation or software is required. But, better to identify problems if they exist than wait to have FDA point them out. It’s important to remember that FDA and other health authorities are sharing their findings, so companies with these problems generally need to deal with multiple regulatory authorities. And those who are contract manufacturers must also address potentially unhappy clients. Remediation of official health authority actions are necessarily more expensive than finding and correcting issues internally.
.webp)

