Reduce Compliance Risk with Real-Time Regulatory Intelligence
Missing a regulatory change or enforcement shift can cost millions in delays, citations, or recalls. Redica gives Regulatory Compliance teams a single, explainable source of truth connecting regulations, inspections, and enforcement trends, so you can act with confidence.


















How Regulatory Compliance Teams Can Use Redica
Continuous Regulatory Monitoring
Maintain awareness of evolving regulations across 50+ countries and 200+ regulatory bodies, guidance updates, and standards.
Global regulatory and guidance monitoring
Topic, GxP, and regional filtering
Draft-to-final change tracking with version history
Explainable AI summaries with source citations
Integrated workflows for impact assessment
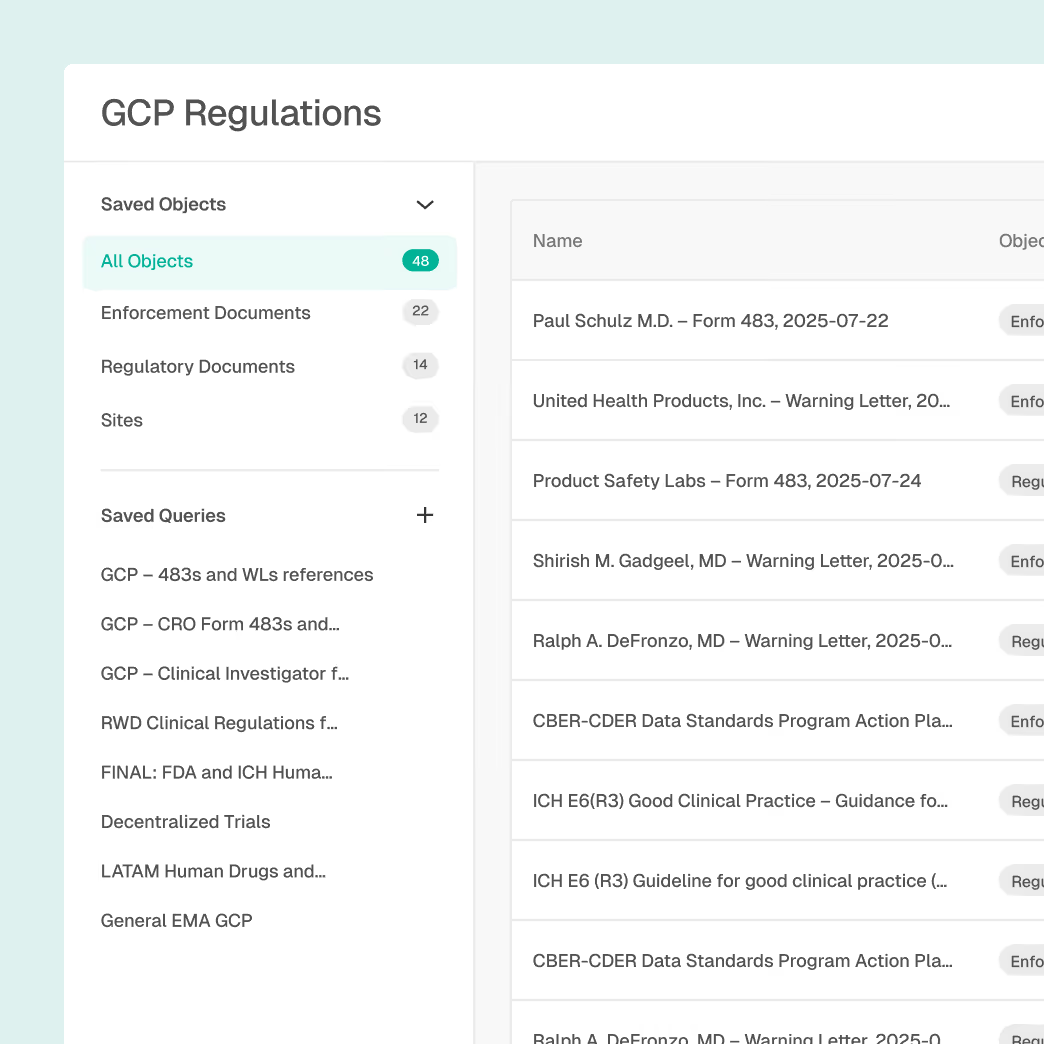
Inspection and Enforcement Oversight
Understand how compliance expectations are enforced in practice.
Global inspection and enforcement intelligence
Trending ciitations and enforcement themes
Agency and regulator focus area insights
Historical context for compliance planning

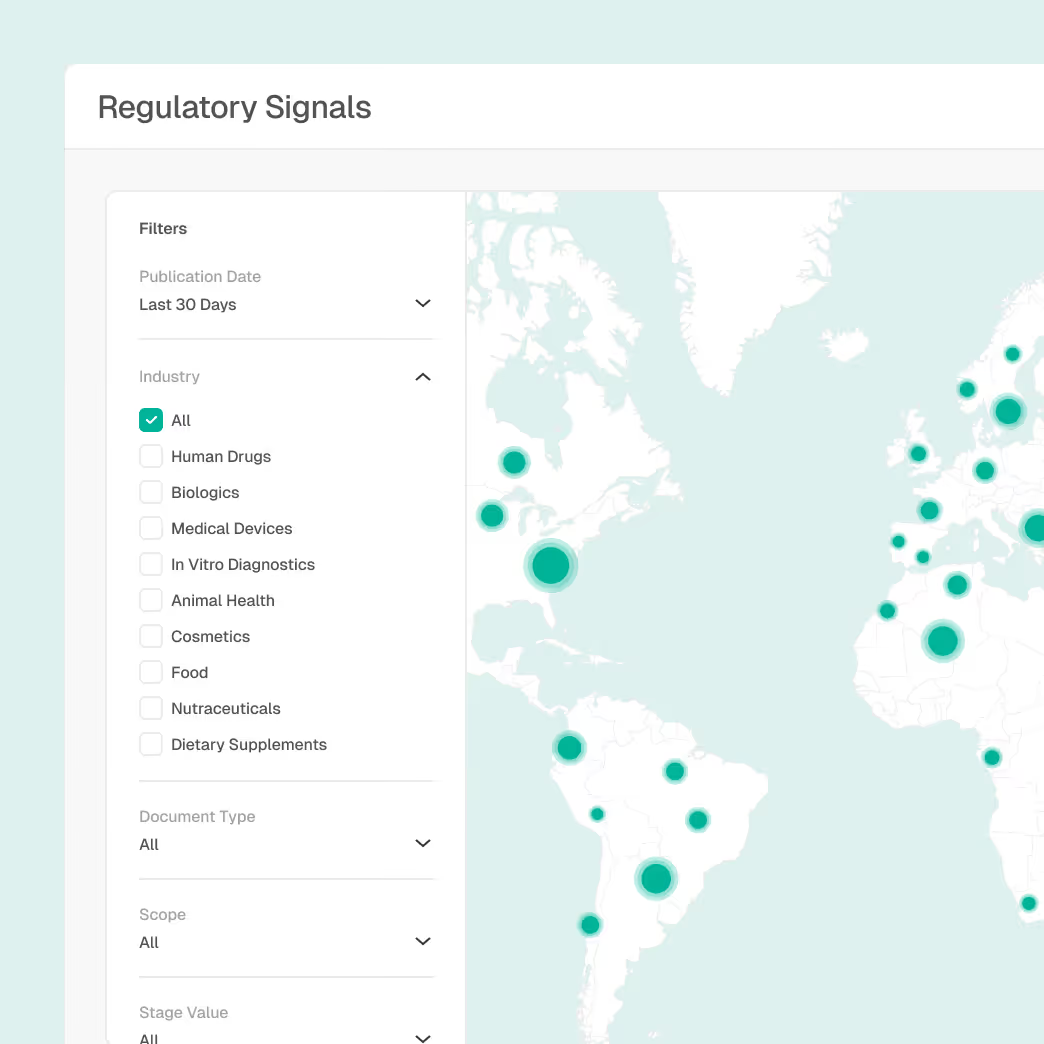
Enterprise Compliance Risk Assessment
Identify and prioritize compliance risk across sites, suppliers, and processes.
Site intelligence with inspection history
AI-assisted detection of emerging risk patterns
Correlation between regulatory updates and enforcement outcomes
Objective, evidence-backed prioritization
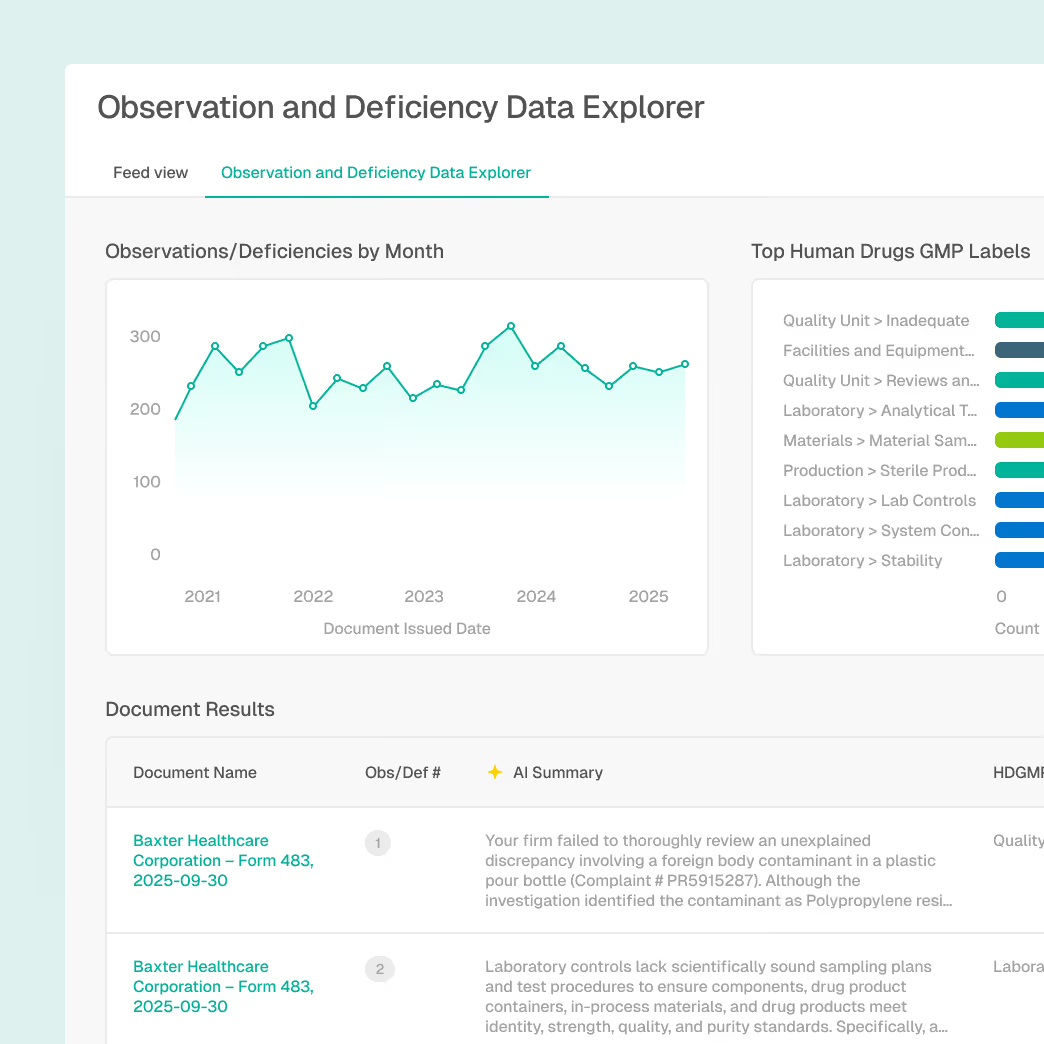
Audit Defense and Compliance Documentation
Support audits and investigations with traceable intelligence.
Centalized access to regulatory and enforcement context
Linked citations and version-controlled sources
Documented rationale for compliance decisions
Explainable AI outputs suitable for audits
What Makes Redica Different
Compliance Grounded in Real Enforcement Behavior
Redica connects regulatory requirements with inspection and enforcement outcomes to show how rules are applied in practice
Explainable Intelligence for Defensible Decisions
Every insight includes citations, provenance, and reasoning steps to support audit-ready compliance.
Built for Global Regulatory Complexity
Powered by the Redica Catalyst Platform and Redica ID, intelligence is standardized across agencies, regions, and regulated activities.
Embedded into Compliance Workflows
Access insights in the Redica App[Link: App] or integrate intelligence into QMS, RIM, and enterprise systems.
.avif)