
Turn Supplier Signals into Supply Risk Intelligence
Supply chain teams need early visibility into supplier risk, site performance, and regulatory enforcement to prevent disruptions. Redica connects regulatory, inspection, site, and post-market intelligence into a single, explainable source of truth to support more resilient, proactive supply chains.


















How Supply Chain Teams Use Redica
Supplier and CMO Risk Management
Identify and monitor risk across suppliers, CMOs, and API manufacturers before issues disrupt supply.
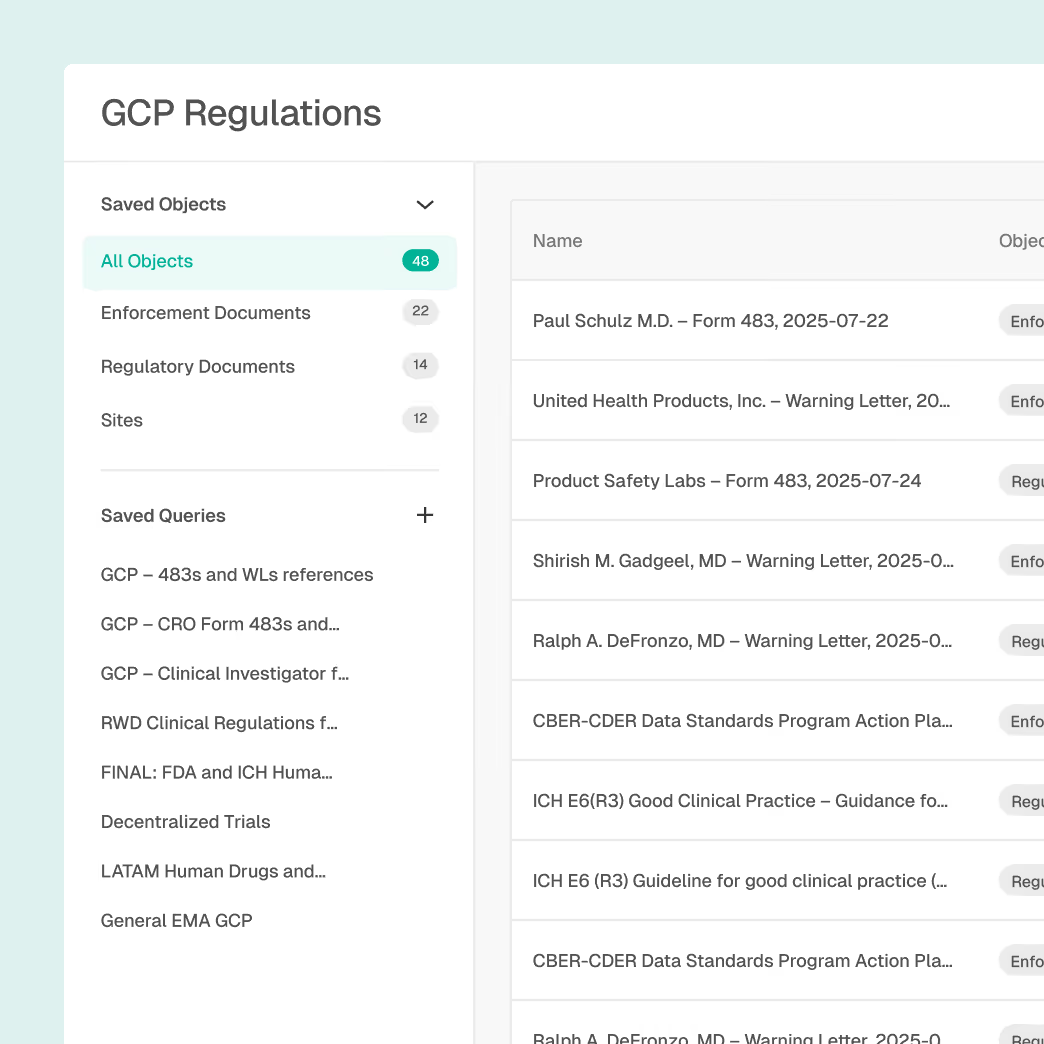
Site Intelligence with global site profiles and inspection history
AI-driven risk scoring based on enforcement trends
Alerts for inspections, warning letters, and compliance actions
Supplier and CMO benchmarking across regions
Early Detection of Supply Disruption Signals
Spot emerging risks that may impact continuity, quality, or availability.
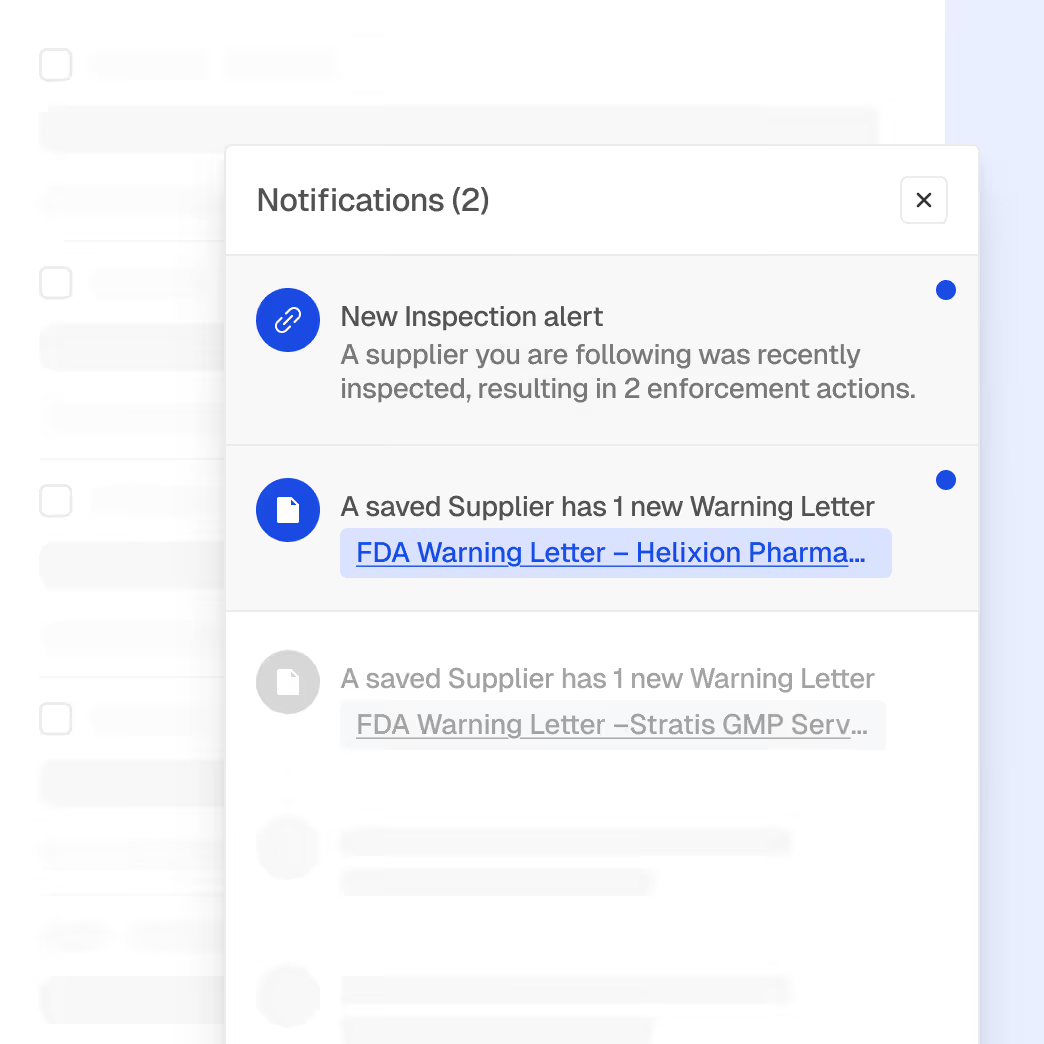
Inspection and enforcement trend analysis
Correlation between site performance and regulatory actions
AI-assisted detection of rising risk indicators
Contextual insights grounded in regulatory behavior
Supplier Qualification and Ongoing Oversight
Support supplier onboarding and periodic review with consistent, objective intelligence.
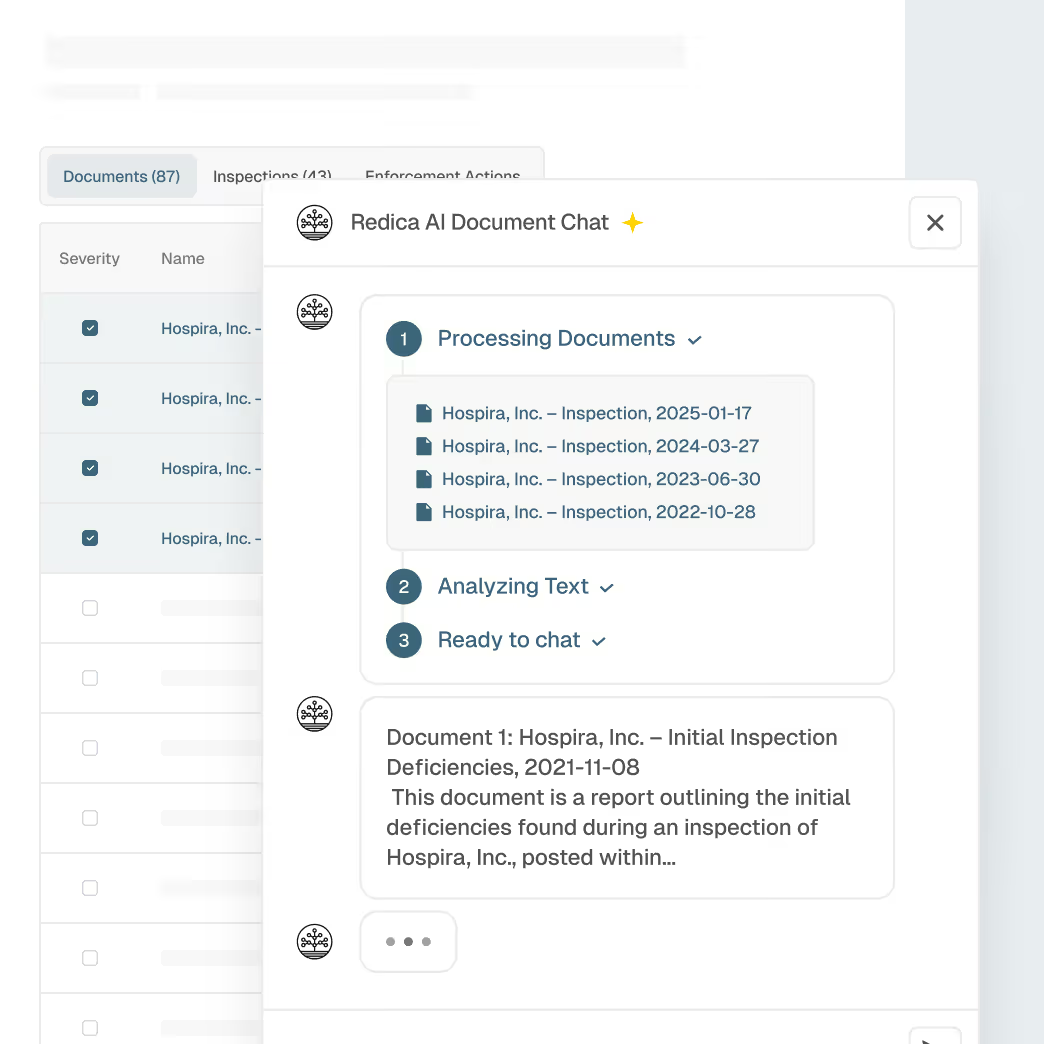
Unified supplier profiles enriched with regulatory and inspection data
Historical performance and enforcement context
Explainable AI summaries for faster assessments
Evidence-backed oversight decisions

Post-Market Signals Informing Supply Risk
Connect downstream product signals to upstream supply chain vulnerabilities.
Recall and quality signal monitoring
Cross-reference with site and supplier inspection history
Unified timeline of events and actions
Faster triage and escalation decisions
What Makes Redica Different
Early Supplier and Site Risk Visibility
Surface regulatory, inspection, and enforcement signals tied to suppliers and manufacturing sites before disruptions occur.
Objective Intelligence for Supplier Decisions
Connect inspection outcomes, warning letters, and performance history to support defensible supplier qualification and oversight.
Built for Global Supply Networks
Standardized supplier and site intelligence across regions, CMOs, and API manufacturers, powered by Redica ID and the Catalyst Platform.
Embedded into. Supply Chain Systems
Access insights in the Redica App or integrate directly into ERP, QMS, and supplier management workflows.
.avif)


