
Unified Data for Enterprise Intelligence
Redica’s data foundation transforms fragmented regulatory, inspection, site, and post-market information into structured, connected, and AI-ready intelligence. Built on the Redica Catalyst Platform, our data is curated, normalized, and continuously enriched to support analytics, AI, and enterprise decision-making with confidence.










Why Most Regulatory Data Fails at Scale
Regulatory and enforcement data is abundant but difficult to use. It spans thousands of global sources, inconsistent formats, unstructured documents, and evolving terminology. Organizations attempting to use this data for analytics or AI often struggle with duplication, incomplete context, unreliable identifiers, and manual data preparation.
Without a strong data foundation, intelligence efforts stall. Analytics become brittle, AI models hallucinate, and teams lose trust in the outputs.
Redica solves this by designing data specifically for intelligence, not just collection.
The Data Foundation Behind the Redica Intelligence Cloud
Curated Global Regulatory and Enforcement Coverage
Redica continuously aggregates regulatory, inspection, and enforcement data from hundreds of global authorities, harmonization bodies, and non-agency sources. Content is ingested daily and refreshed continuously to reflect evolving regulatory and enforcement landscapes.
.png)
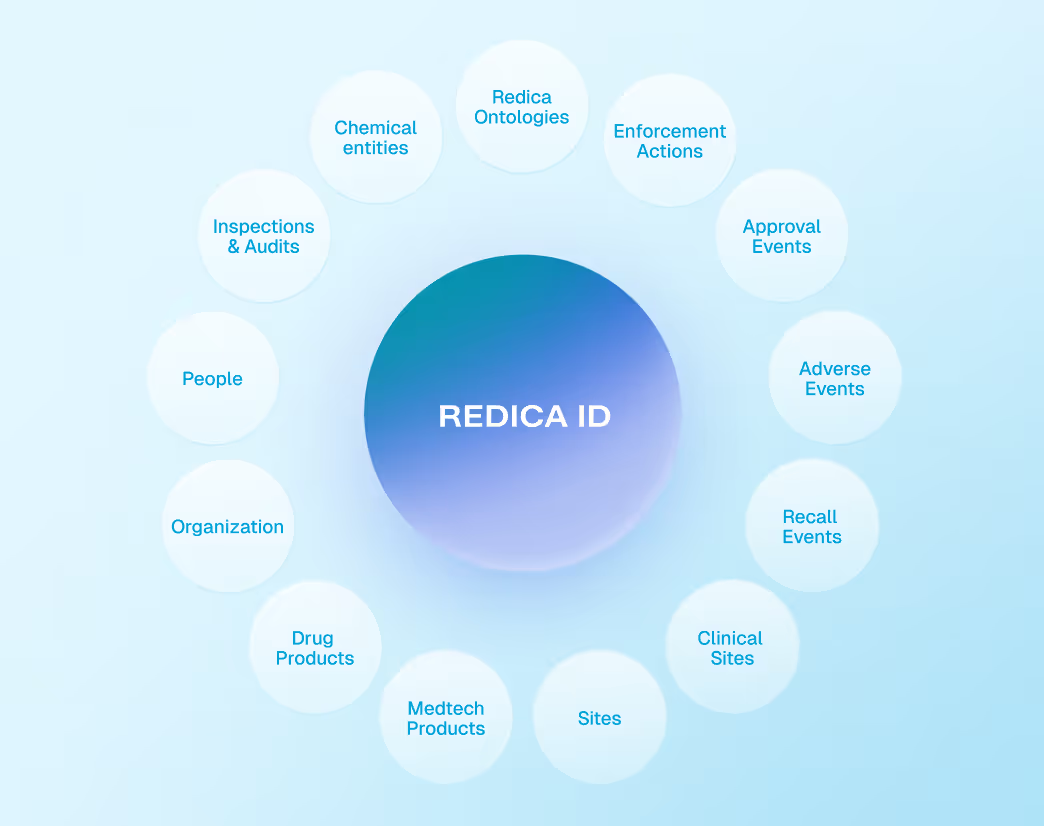
Redica ID for Entity Resolution and Consistency
Redica ID resolves sites, suppliers, products, organizations, and regulators into consistent identities across all datasets. This ensures accurate linking, benchmarking, and analysis across regions, systems, and data sources.
DocIQ for Structured Intelligence from Documents
DocIQ parses, translates, and structures complex regulatory and enforcement documents, including scanned PDFs and non-English content. Every document is summarized, labeled, and linked to entities and topics for reliable downstream use.

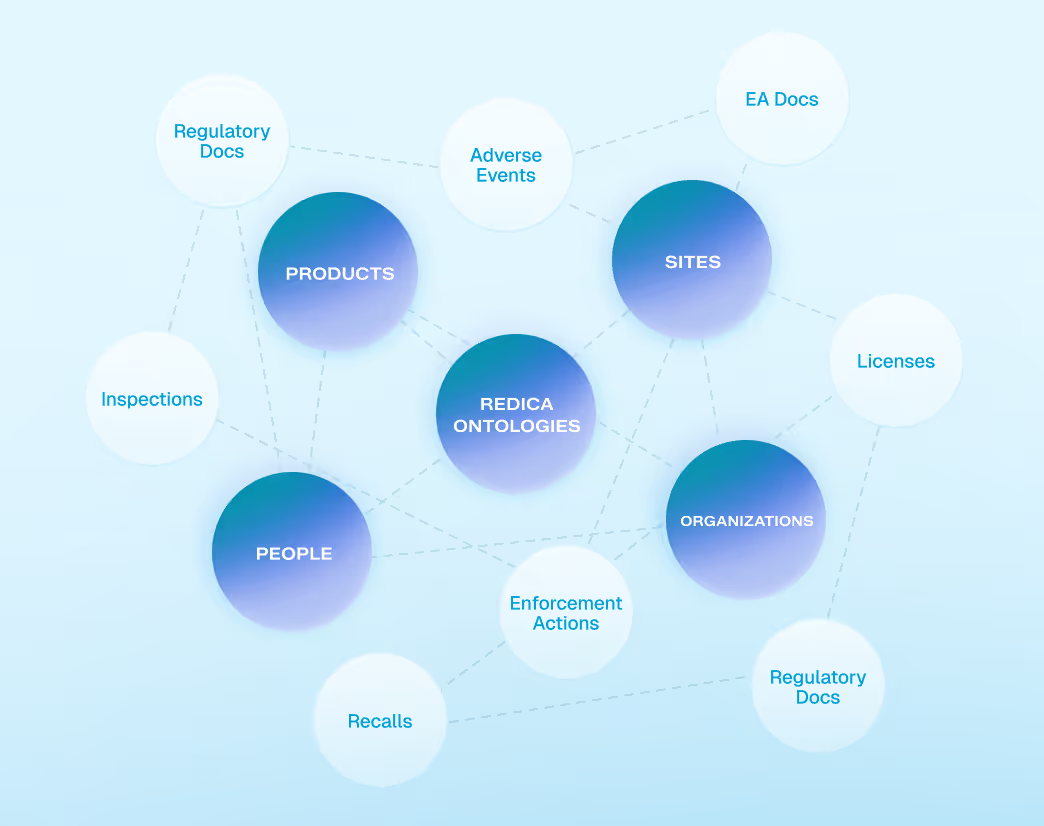
Knowledge Graph for Context and Relationships
Redica’s knowledge graph encodes relationships between regulations, inspections, sites, products, and enforcement actions. This enables contextual analysis, trend detection, and AI reasoning beyond simple keyword or table-based approaches.
AI-Ready Data Built on the Catalyst Platform
All Redica data is standardized, labeled, and enriched on the Redica Catalyst Platform. This foundation supports analytics, machine learning, and advanced AI workflows with full traceability and governance.
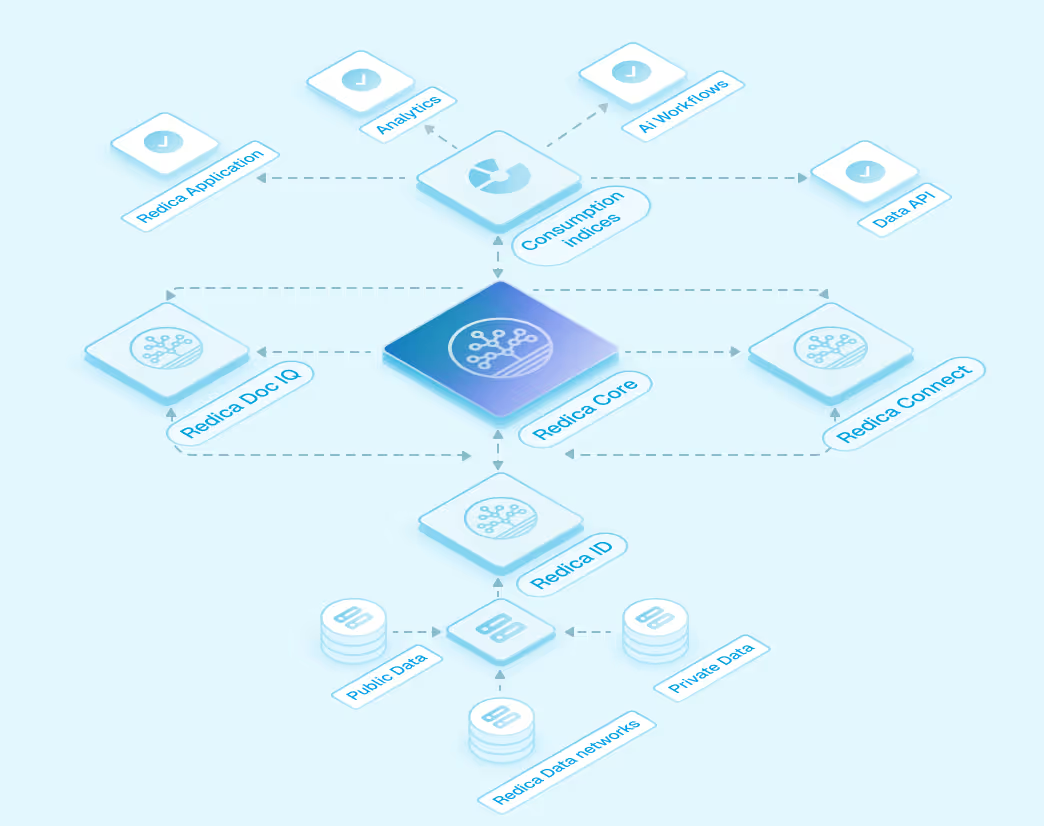
How Teams Use Redica Data
Analytics and Benchmarking
Compare performance across sites, suppliers, regions, and product types using consistent, normalized datasets.
AI and Machine Learning Enablement
Train and deploy models using structured, labeled, and explainable data grounded in authoritative regulatory and enforcement sources.
Enterprise Intelligence and Decision Support
Power dashboards, reports, and decision workflows with connected data that reflects real-world regulatory behavior.
Redica Data by the Numbers
regulators, standards bodies, and trade associations monitored
global content ingestion and change detection
precision rate in labeling and reviewing document signals
version history maintained for new documents
translation of all non-English documents
years of regulatory and inspection history
compliant data infrastructure
FAQ
How does Redica ensure data quality and accuracy?
Data is curated using automated pipelines combined with human-in-the-loop review, version tracking, and continuous validation.
How does Redica prevent bad data from undermining analytics and AI?
Redica data is designed for intelligence from the start. Every record is normalized, labeled, versioned, and linked using Redica ID, DocIQ, and the knowledge graph, with human-in-the-loop review and full provenance. This ensures analytics and AI workflows are grounded in consistent, explainable data rather than brittle or incomplete inputs.
How is Redica data structured differently from raw feeds?
All data is normalized, labeled, and linked using Redica ID, DocIQ, and the Redica knowledge graph to ensure consistency and usability.
Is the data suitable for analytics and AI?
Yes. Redica data is purpose-built for analytics and AI, with structured schemas, semantic labels, and traceable provenance.
What types of data does Redica provide?
Redica covers regulatory guidance, inspection and enforcement records, site profiles, post-market signals, and related metadata across global jurisdictions.
Still have questions? Contact us


