
Stay Ahead with Connected Regulatory Intelligence
Redica delivers proactive, contextual, AI-enriched insights so regulatory, quality, and compliance teams can anticipate regulatory changes, assess their impact, and take action where and when it counts.










Streamlined, Scalable Compliance
Traditional monitoring tools only show you what changed, not why it matters, or how it impacts your SOPs, filings, or quality processes.
In a landscape where regulators issue hundreds of new guidances and updates monthly, compliance is challenged by disconnected feeds, manual tracking, and siloed systems.
Redica AI tracks individual regulations with precision to transform complex regulatory shifts into trusted, relevant insights with integrated workflows for faster, smarter compliance.
Focused Intelligence to Unravel Regulatory Complexity
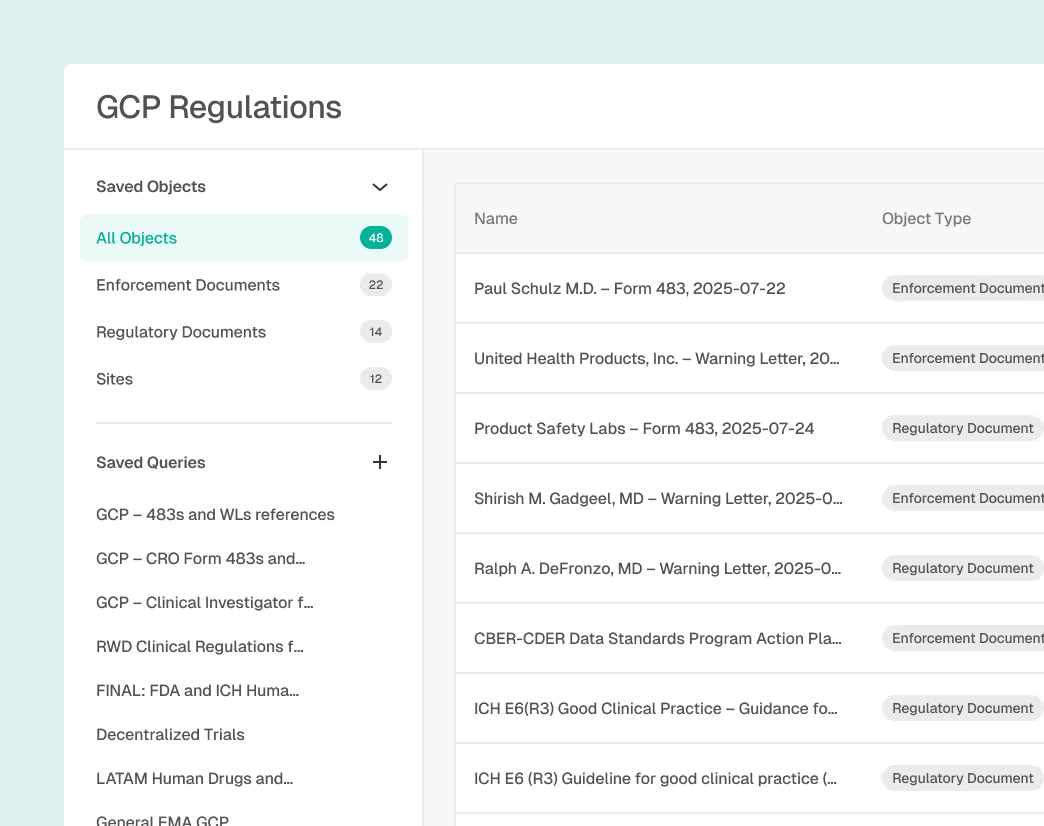
Unified Global Regulatory Data
Access connected regulatory updates and enforcement data in one place to immediately understand what changed and why it matters.
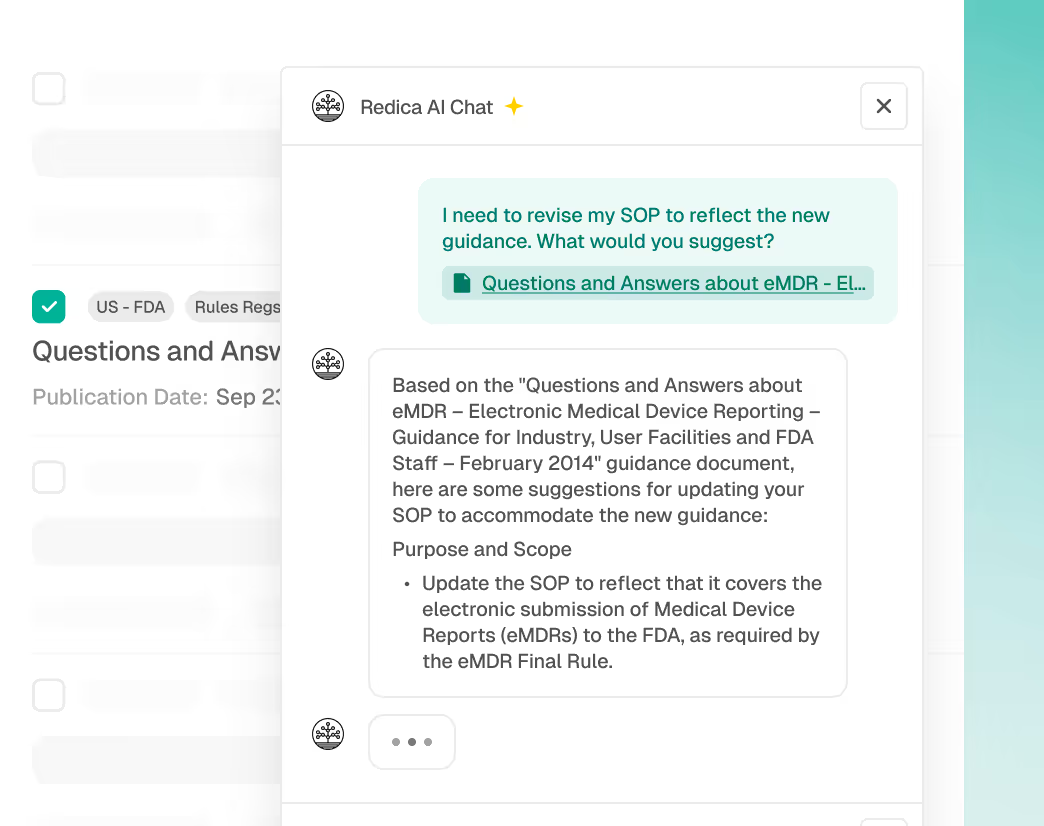
From Signal to Action
Turn regulatory alerts into clear, actionable recommendations with AI copilots that help summarize and quantify regulatory impact, identify impacted divisions and subject matter experts, identify documents and processes for updates, and accelerate SOP and other documentation updates.
Impact Assessment AI Workflows
Automatically map regulatory change to SOPs, submissions, CAPAs and assign and define impact across your global network with human in the loop processes.


Explainable, Audit-Ready Intelligence
Trace every piece of intelligence and recommendation to the original source and logic so you have full evidence and traceability.
Embedded Intelligence with Veeva and QMS Integrations
Work in the Redica app, integrate intelligence directly into Veeva RIMS, Quality Docs, and QMS workflows, or leverage Redica's integrated solutions that process and connect your internal company data for more holistic insights and automation.
Regulatory Intelligence for Every Function
Track Draft Guidance and Final Rule Changes
Monitor regulatory changes with granularity as they move from draft to final, add commentary, compare versions, and route updates to relevant internal teams.
Filter and Prioritize Signals
Labels by GxP, topic, product type, MedTech code, process, region, and impact level allow teams to immediately focus only on material changes.
Connect Regulatory Updates to Enforcement Behavior
Cut through the noise with a unified, curated view of contextually relevant global regulatory changes that correlates updates with citations, inspection narratives, and warning letters.
With Redica, I can leave the noise behind.
Dawn Carlson
Vice President, Global Compliance and Regulatory Affairs

Discover Regulatory Excellence with Redica
Regulators, standards bodies, and trade associations monitored
Labels for faster filtering and data discovery
Global regulatory content ingestion & change detection
Connected version history for all new documents
Publish of core US regulatory signals
Refreshed sources annually for proactive, relevant change monitoring
Regulatory documents published monthly
FAQ
Can it integrate with our systems?
Yes. Redica delivers data and intelligence through the Redica app and via API for integrations with other solutions like Veeva RIM, Veeva Quality Docs, QMS, Snowflake, and document management systems.
Does it support AI explainability?
All AI-driven summaries are complete with data provenance, including source citations and reasoning steps for full audit traceability.
Does Redica provide enforcement intelligence alongside regulatory updates?
Yes. Redica uniquely unifies regulatory change with enforcement trends, citations, and inspection narratives for proactive risk forecasting.
How often is the data updated?
Continuously. Redica aggregates and enriches data from hundreds of global health authorities and standards bodies with the most trusted, current, and comprehensive source for regulatory intelligence.
What makes Redica different from other monitoring tools?
Most systems surface new regulations. Redica tracks, classifies, and connects regulatory data by topic, industry, scope, product, and regulatory area, and seamlessly routes only contextually relevant rules, regulations, and guidance to you for impact assessment for your SOPs, products, and submissions so you can act immediately.
Still have questions? Contact us



