
Navigate Global Regulatory Change with Confidence
Regulatory Affairs teams need clarity across evolving regulations, enforcement trends, and product impact. Redica connects regulatory, inspection, site, and post-market intelligence into a single, explainable source of truth to support faster, more defensible regulatory decisions.


















How Regulatory Affairs Teams Use Redica
Global Regulatory Change Monitoring
Stay ahead of evolving regulations, guidance, and standards across regions and agencies.
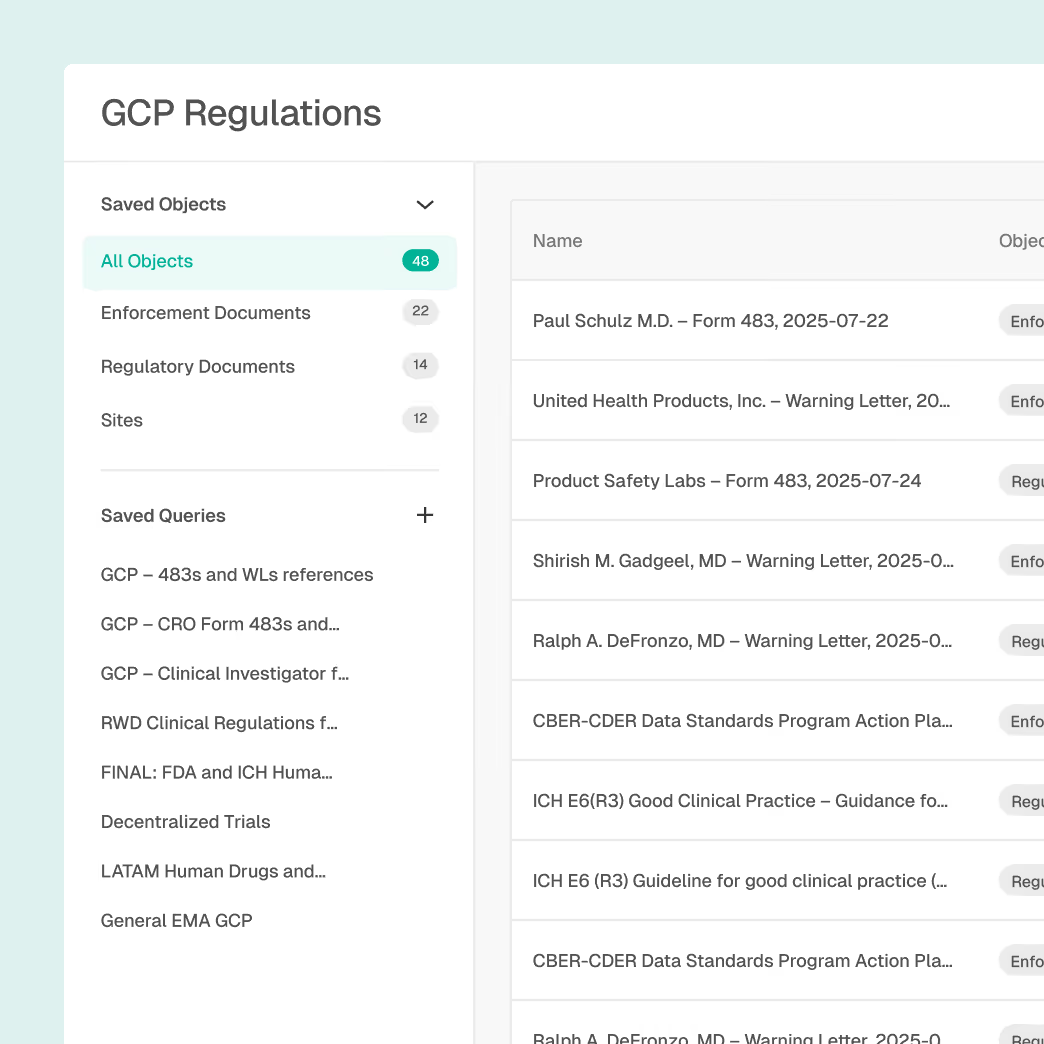
Regulatory Intelligence with global coverage
Draft-to-final tracking with version history
Topic and GxP-based filtering
Explainable AI summaries with source citations
Regulatory Impact Assessment & Change Control
Understand what regulatory changes mean for your products, submissions, and documentation.
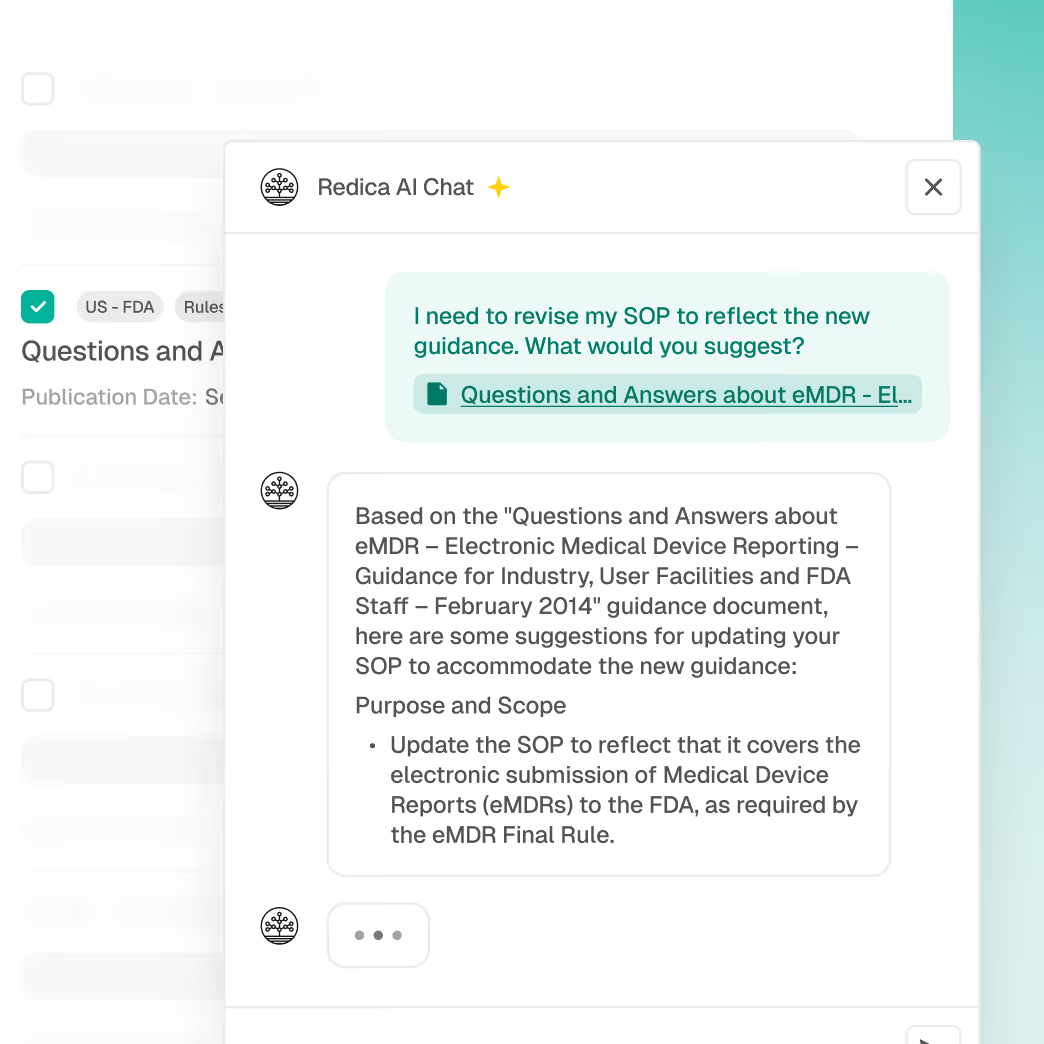
Automated mapping of updates to SOPs and processes
Product, indication, and market-level impact analysis
AI-assisted summaries for faster assessments
Traceable evidence for audit defense
Inspection & Enforcement Context for Regulatory Strategy
Inform regulatory strategy with insight into how agencies enforce requirements in practice.
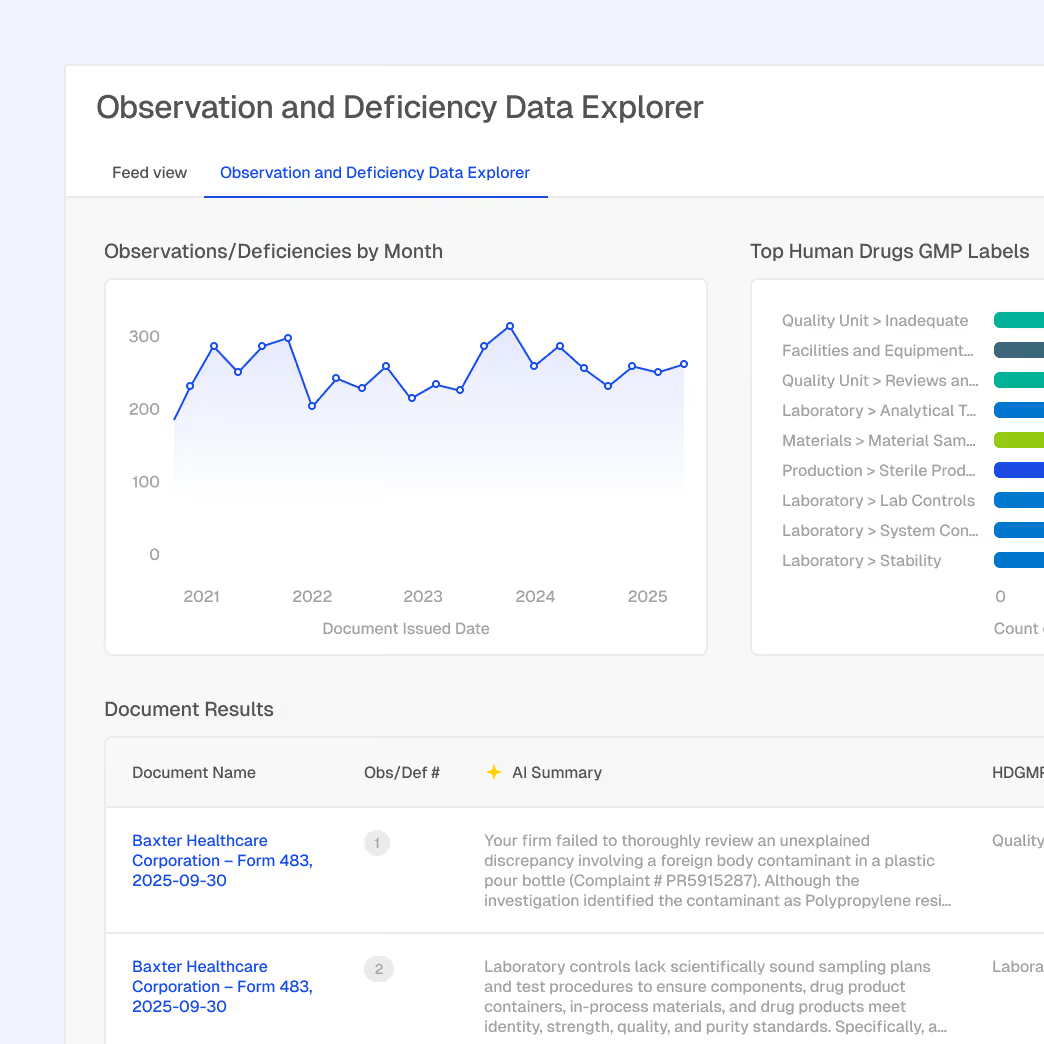
Inspection and enforcement trend analysis
Correlation between guidance updates and citations
Regulator and agency focus area insights
Unified view of policy and enforcement behavior
Submission & Portfolio Oversight
Support consistent regulatory decision making across products and markets.
Centralized view of regulatory signals by product line
Cross-market visibility into requirements and expectations
Early signals that may impact filings or variations
Connected intelligence across lifecycle stages

What Makes Redica Different
Regulatory Intelligence with Real-World Context
Redica connects regulatory updates with inspection and enforcement behavior so teams understand not just what changed, but how it is applied.
Explainable, Audit-Ready Insights
Every insight includes citations, version history, and reasoning steps to support defensible regulatory decisions.
Built for Global Regulatory Complexity
Powered by the Redica Catalyst Platform and Redica ID, intelligence is standardized across agencies, regions, and product portfolios.
Embedded into Regulatory Workflows
Access insights in the Redica App or integrate directly into systems of record, document management, and enterprise systems.
.avif)


