
Site Intelligence Cloud:
One Platform. Every Signal.
The only platform that unites inspection data from 125+ global health authorities with AI-powered summaries and leading risk indicators into a cloud-level overview of global site and supplier risk.
Quality, Supplier Management, and Compliance teams can now
replace fragmented manual searches with proactive supply chain risk intelligence to detect emerging issues faster than ever before.









The Hidden Cost of
Fragmented Data
Supplier networks are expanding, manufacturing models are more distributed, and regulatory scrutiny of third-party quality practices continues to rise. Quality teams spend 80% of their time gathering data, and only 20% acting on it. Scattered documents, inconsistent audits, and periodic reviews often reveal issues only after escalation, leaving pharma supply chain risks undetected until it's too late.
Site Intelligence Cloud flips that ratio! Redica unifies global site, supplier, regulatory, and inspection data into a single composite risk score. That score is continuously updated, AI-powered, and designed to surface supply chain risk earlier and with deeper context than any tool built for a single data source.
Focused Intelligence for Supplier and Site Oversight
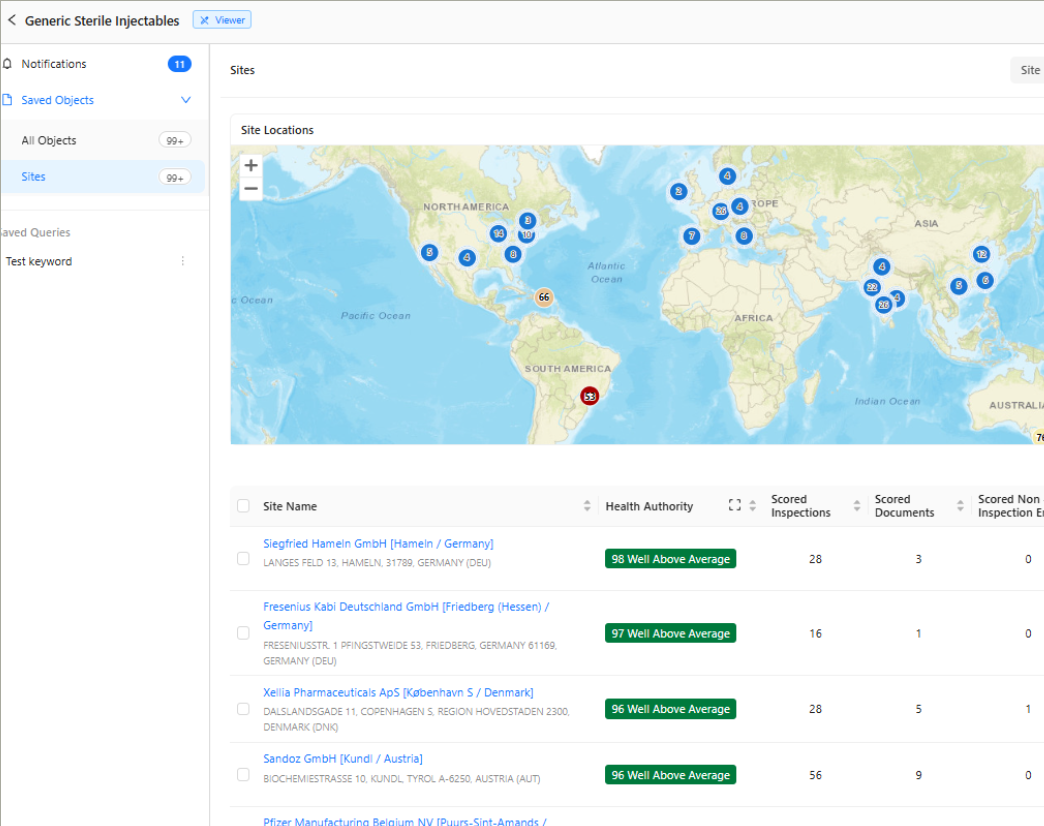
Unified Global Site & Supplier Risk Intelligence
View regulatory actions, inspection history, warning letters, adverse trends, and operational indicators in one connected site profile. A composite risk score drawn from 14 scored global health authorities replaces manual research with objective and up-to-date intelligence for every site you monitor.

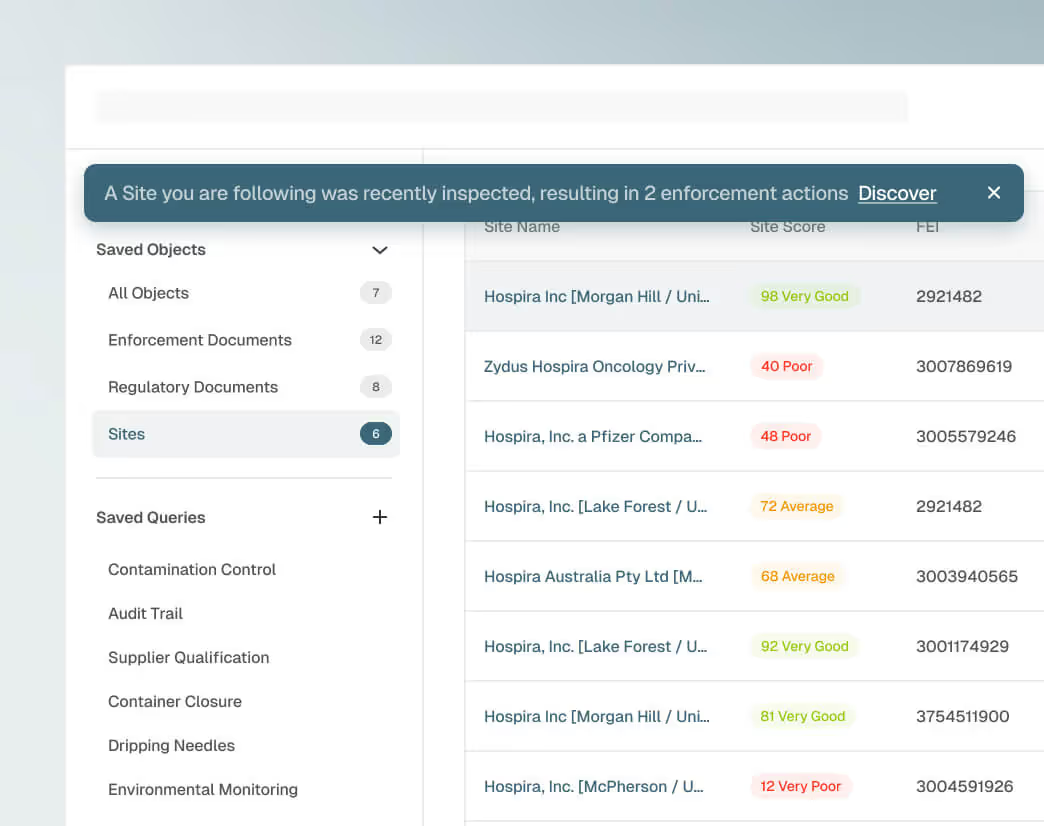
Early Signals for Risk Detection
Get ahead of warning signs at suppliers and CMOs before they escalate. Forward-looking risk indicators surface risk weeks or months ahead of action. Now your team can prioritize resources and intervene early with evidence.
Comprehensive Site Profiles with Contextual Insights
Access manufacturing sites, contract manufacturers, and critical partner profiles with AI-enriched enforcement patterns, site behavior, historical citations, and cross-site comparisons. AI risk summaries explain what’s driving each score in plain language, giving your team the full picture at a glance.
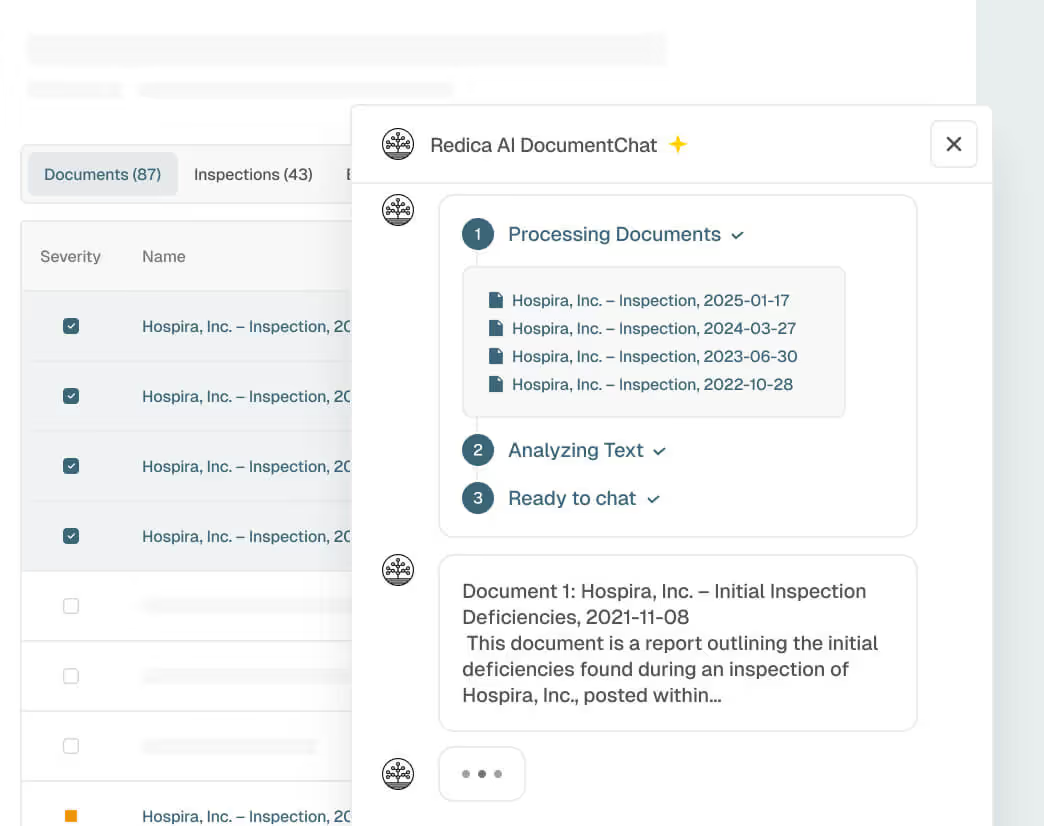
Explainable, Audit-Ready Intelligence
Every risk indicator, AI-generated recommendation, and site classification includes transparent provenance and logic to support audits, supplier qualification, and risk mitigation.
Embedded Intelligence with QMS, ERP, and Supply Chain Integrations
Work in the Redica app, integrate intelligence directly into QMS workflows, or leverage Redica integrated solutions that process and connect your internal company data for more streamlined, holistic oversight.
Use Site Intelligence directly in the Redica App or embed insights into supplier quality, procurement, QMS, and ERP workflows. Redica integrated solutions combine internal performance data with external risk signals for a holistic risk view.
Redica integrated solutions combine your internal quality data with external regulatory intelligence for a unified picture.
Stronger Oversight from
Quality to Procurement
Strengthen Supplier Qualification & Monitoring
Evaluate new and existing suppliers with AI-enriched inspection history, enforcement signals, and risk indicators to support reliable qualification and continuous monitoring. Standardized scoring replaces subjective decisions with objective, auditable data your team can defend and stand behind.
Prioritize Oversight Using Emerging Risk Signals
Identify suppliers or sites requiring additional audits, CAPAs, or escalation based on early warning signals and regulator activity. Leading Risk Indicators surface Warning Letter and CRL risk weeks or months before enforcement, giving you mitigation time before a disruption, not after.
Benchmark, Qualify, and Onboard with Confidence
Use comprehensive site profiles and objective benchmarking to support sourcing strategies, risk reviews, and cross-functional decision-making. Site Intelligence Cloud extends beyond Quality. Supplier onboarding, M&A due diligence, and CDMO evaluation all run on the same objective scoring and benchmarking capabilities.
This is a powerful analytics tool we use routinely across the full GxP spectrum to monitor supply chain risks, assess vendors, and clinical trial sites. The analytics allow for effective and efficient compliance benchmarking and QMS planning in an easy-to-use format.
Vice President, Global Head of Quality

The Numbers Behind the Intelligence
Global site profiles with inspection history across GMP, GLP, GCP, and GDP
Global regulators and health authorities covered
Years of regulatory & inspection history
For enforcement prediction
Scored health authority data sources
Documents summarized with Redica AI
FAQ
Can it integrate with our systems?
Yes. Site Intelligence can be accessed via the Redica App or embedded into QMS, ERP, procurement, supplier management, or Snowflake environments.
Does it support AI explainability?
All AI-driven indicators and recommendations include transparent logic and source citations for audit-ready traceability.
Does Redica help identify suppliers that need immediate attention?
Yes. AI-powered risk detection highlights suppliers or sites demonstrating rising violations, enforcement flags, or emerging issues that require closer oversight.
How often is the data updated?
Continuously. Redica AI agents ingest and enrich inspection records, enforcement actions, regulatory updates, and other external signals tied to global sites.
What makes Redica different from other supplier risk tools?
Most tools rely solely on self-reported or internal data. Redica AI unifies global inspection, enforcement, regulatory, and operational data to reveal emerging supplier and site risks earlier and with deeper context.
Still have questions? Contact us



