
Advance Pharma Quality and Compliance with Connected Intelligence
Pharmaceutical organizations need complete visibility across regulatory change, inspections, suppliers, and post-market signals to maintain quality and ensure global compliance. Redica unifies these intelligence layers into a single, explainable source of truth for actionable insights across the product lifecycle.


















How Pharma Teams Use Redica
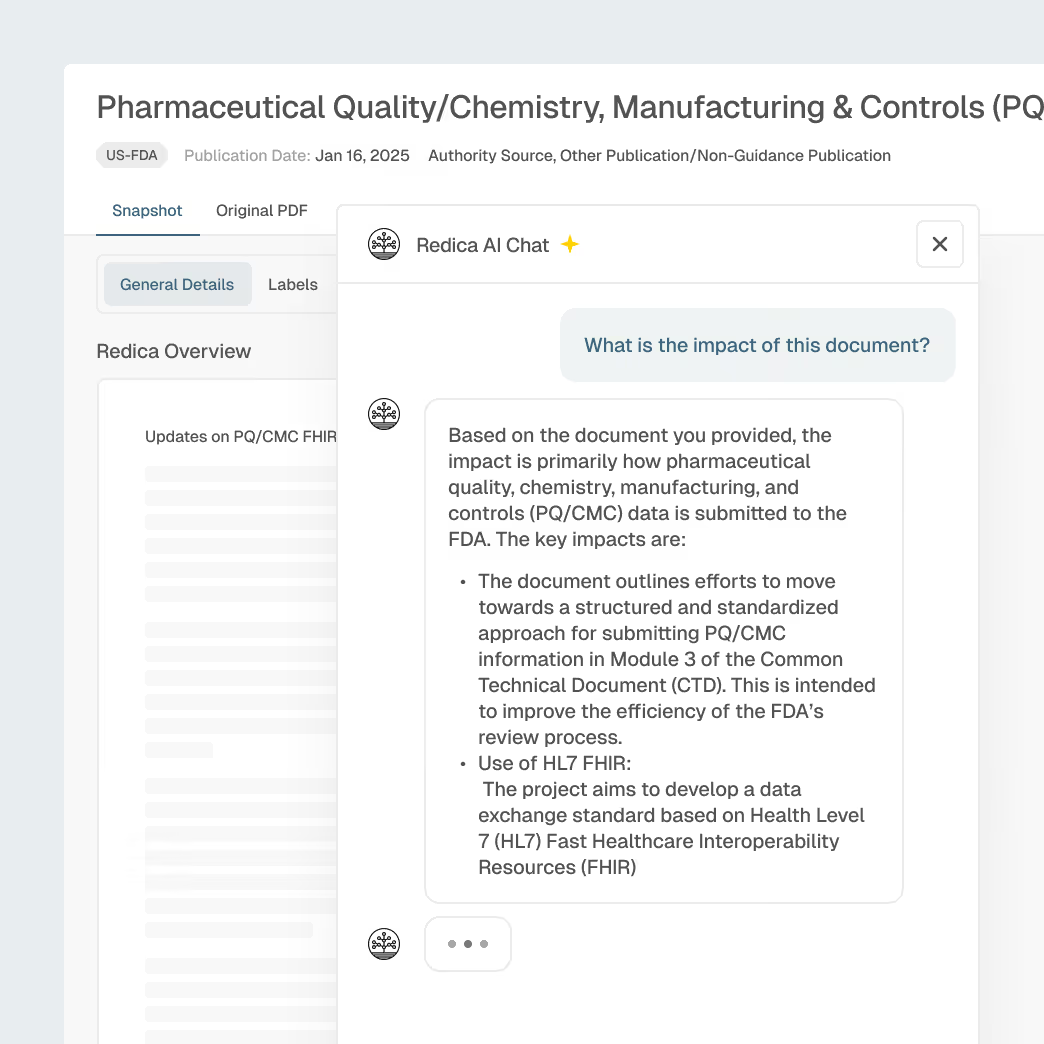
Regulatory & CMC Change Monitoring
Manage evolving global regulations, guidance updates, and CMC expectations with timely AI-enriched insights.
Global regulatory and guideline monitoring
Topic and GxP-based filtering to identify relevant changes
AI summaries for submission and CMC document impact
SOP and process impact linking for faster change control
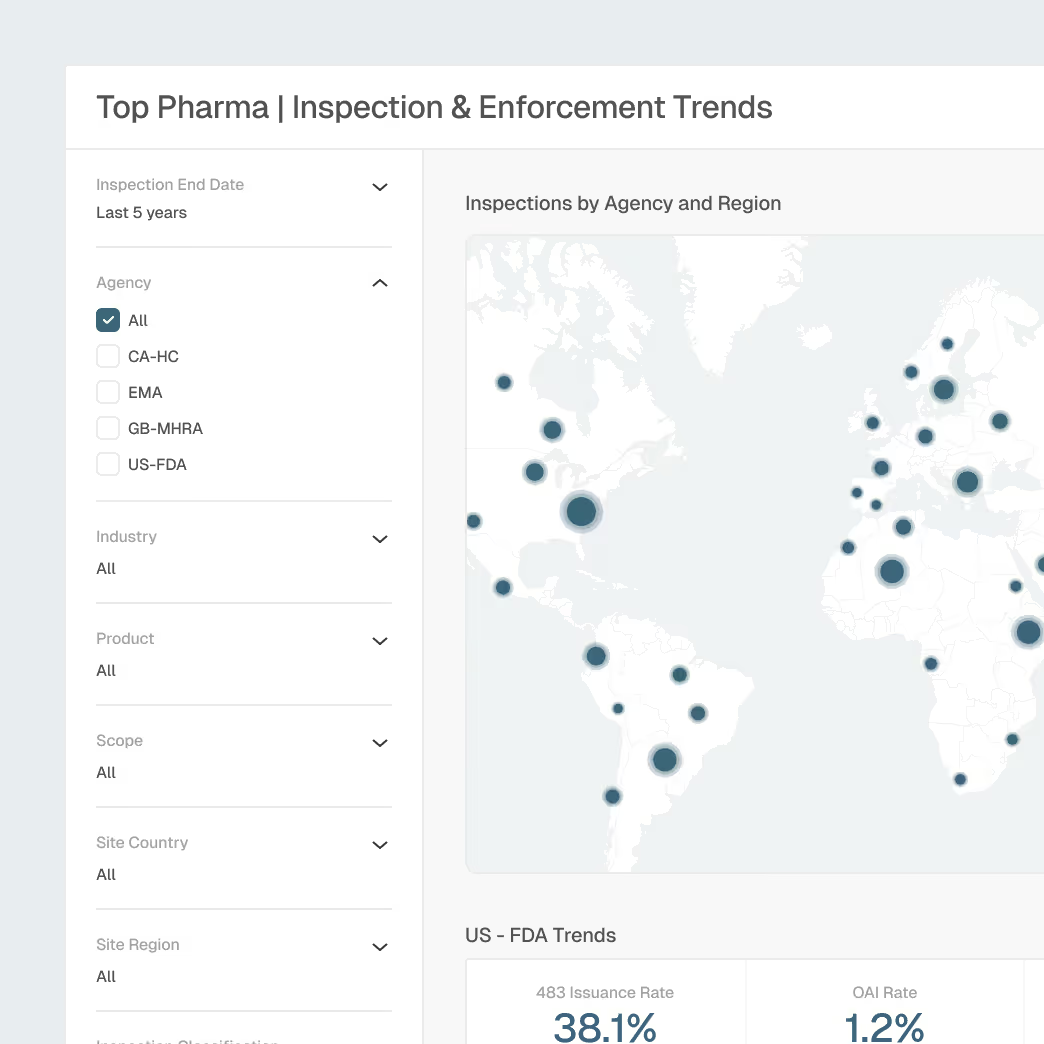
Inspection Readiness Across Global Sites
Prepare for agency inspections armed with insights into regulator focus areas, historical citations, and emerging trends.
Global inspection and enforcement intelligence
Inspector, firm, and site behavioral profiles
Trending issue analysis by product type, region, and agency
Explainable AI summaries for rapid site preparation
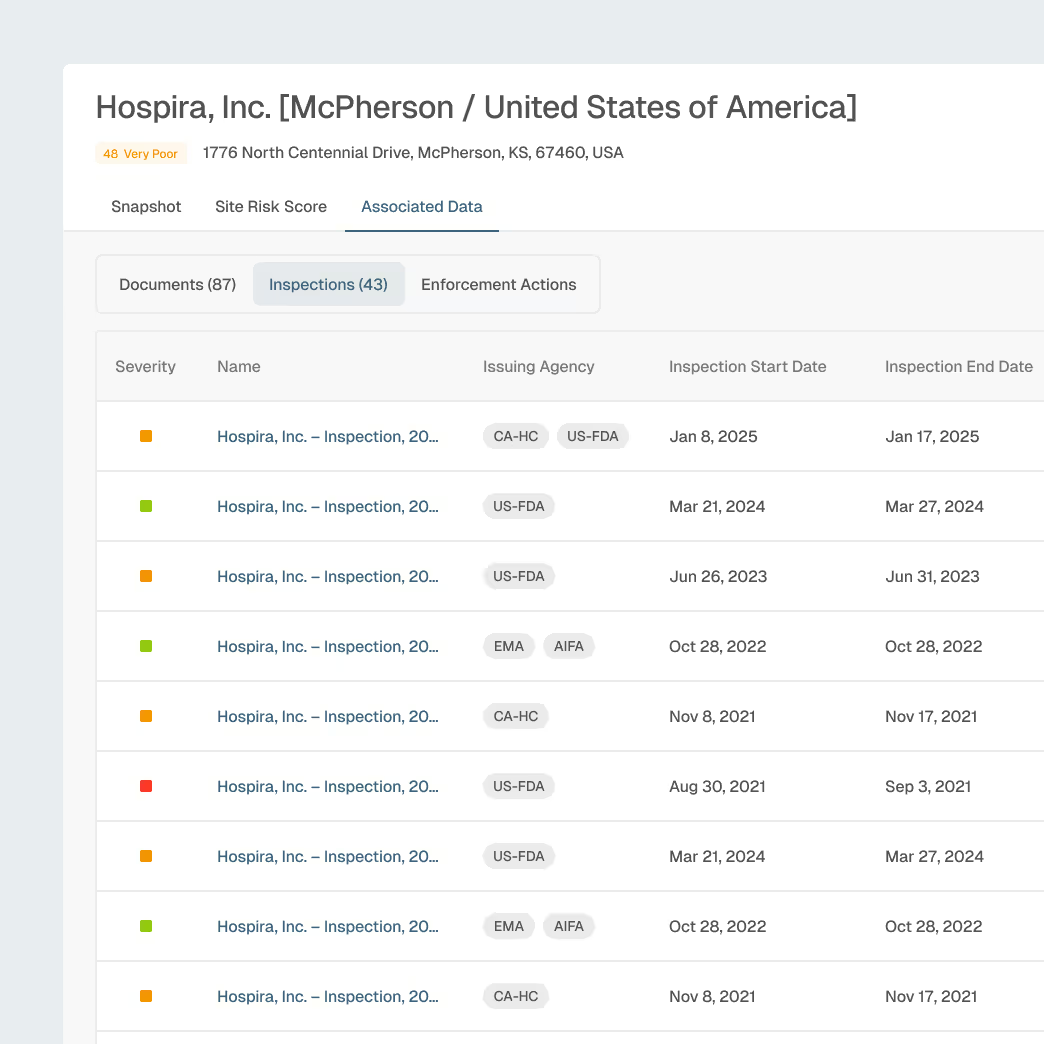
Supplier, CMO, and API Oversight
Strengthen oversight across CMOs, API manufacturers, and third-party partners with unified visibility into site-level risk.
Comprehensive site and supplier profiles
AI-detected early signals of quality or compliance risk
Benchmarking to evaluate supplier reliability
Focused alerts for inspections, warming letters, or enforcement activity
Post-Market Quality & Safety Surveillance
Identify emerging product risk by connecting global safety, recall, and quality signals to inspection and regulatory context.
Adverse event, recall, and safety trend monitoring
AI clustering of product and market signals
Cross-reference against enforcement patterns
Unified timeline for rapid signal triage
.png)
Why Pharma Companies Choose Redica
End-to-End Lifecycle Intelligence
Redica connects regulatory, inspection, supplier, and post-market intelligence so teams can monitor risk and respond with full context.
Explainable AI for Confident Decisions
Every insight includes citations, provenance, and reasoning steps, ensuring decisions stand up to internal and external scrutiny.
Built for Global Pharma Complexity
Powered by Redica Catalyst and Redica ID, intelligence is standardized across sites, markets, and product portfolios.
Embedded Info Pharma Workflows
Access insights in the Redica App or embed intelligence directly into QMS, RIM, ERP, and supply chain systems for seamless execution.
.avif)


