
Manage Cosmetics Compliance in a Rapidly Evolving Landscape
Cosmetics companies face increasing regulatory scrutiny, evolving ingredient requirements, and heightened enforcement around labeling and claims. Redica connects regulatory, inspection, site, and downstream signals into a single, explainable source of truth to help teams detect risk early and respond with confidence across global markets.


















How Cosmetics Companies Use Redica
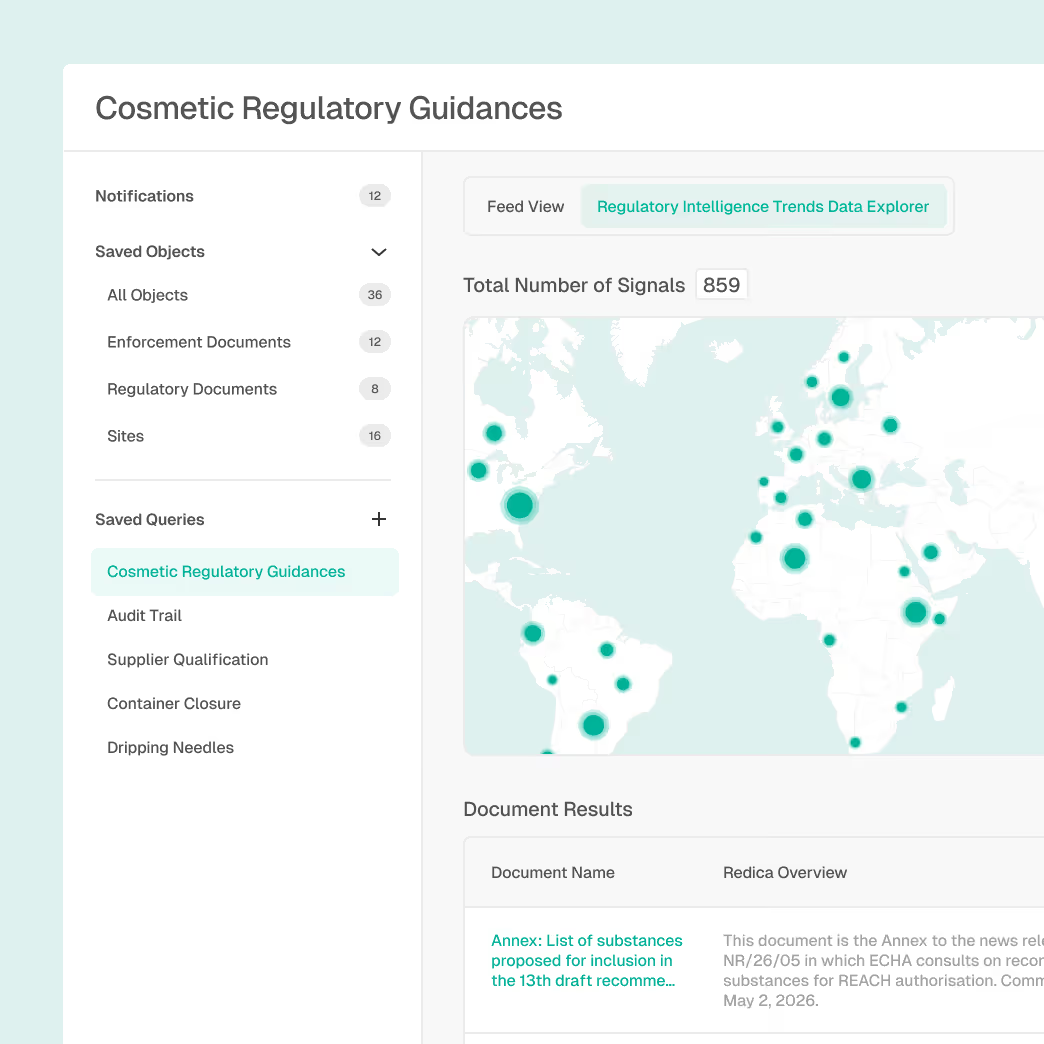
Regulatory Monitoring
Stay ahead of changing cosmetic regulations, ingredient restrictions, and regional requirements.
Global monitoring of FDA cosmetic regulations and guidance
Warning letter and enforcement action tracking
Draft-to-final regulatory change tracking with version history
Explainable AI summaries with traceable citations
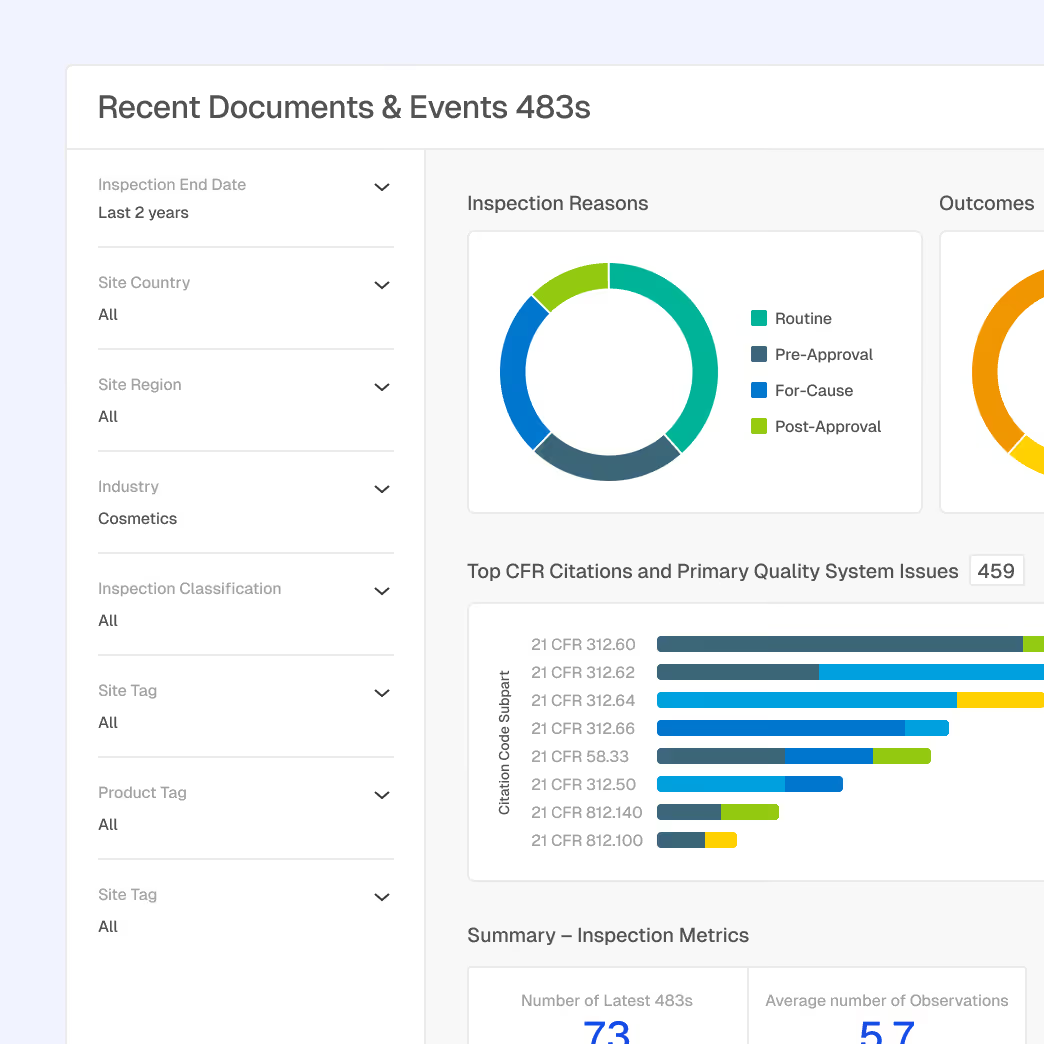
Enforcement Pattern Analysis
Learn from FDA enforcement actions to identify compliance risks before they result in citations or warning letters.
FDA warning letter analysis for adulteration, misbranding, and manufacturing quality violations
Trending citation patterns from Form 483 observations across cosmetics sites
Enforcement theme detection
Historical enforcement context for internal compliance prioritization
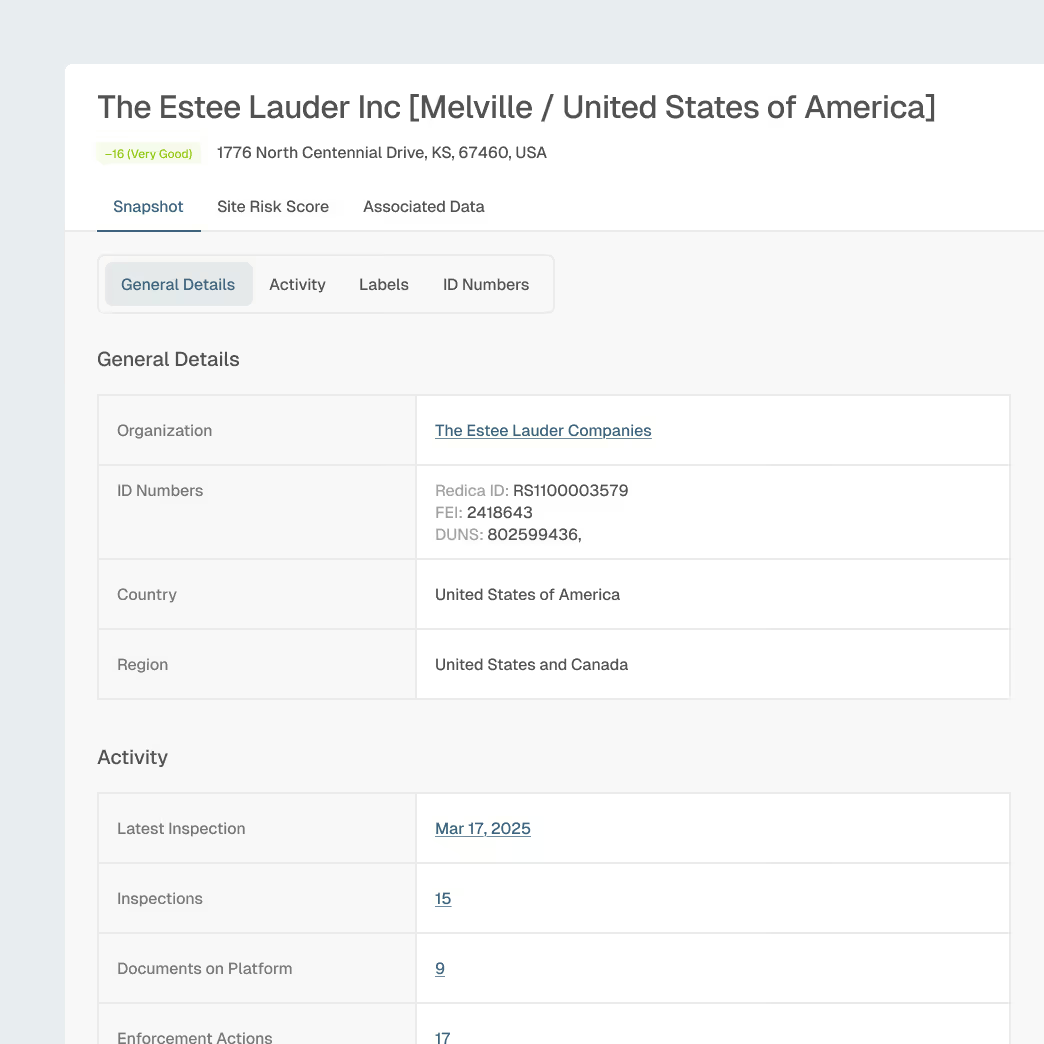
Manufacturing and Facility Intelligence
Understand FDA inspection outcomes across cosmetics manufacturing sites.
Site-level FDA inspection history (Form 483 observations)
Warning letter tracking tied to specific firms and sites
Compliance patterns across manufacturing locations
AI-assisted supplier evaluation and audit planning
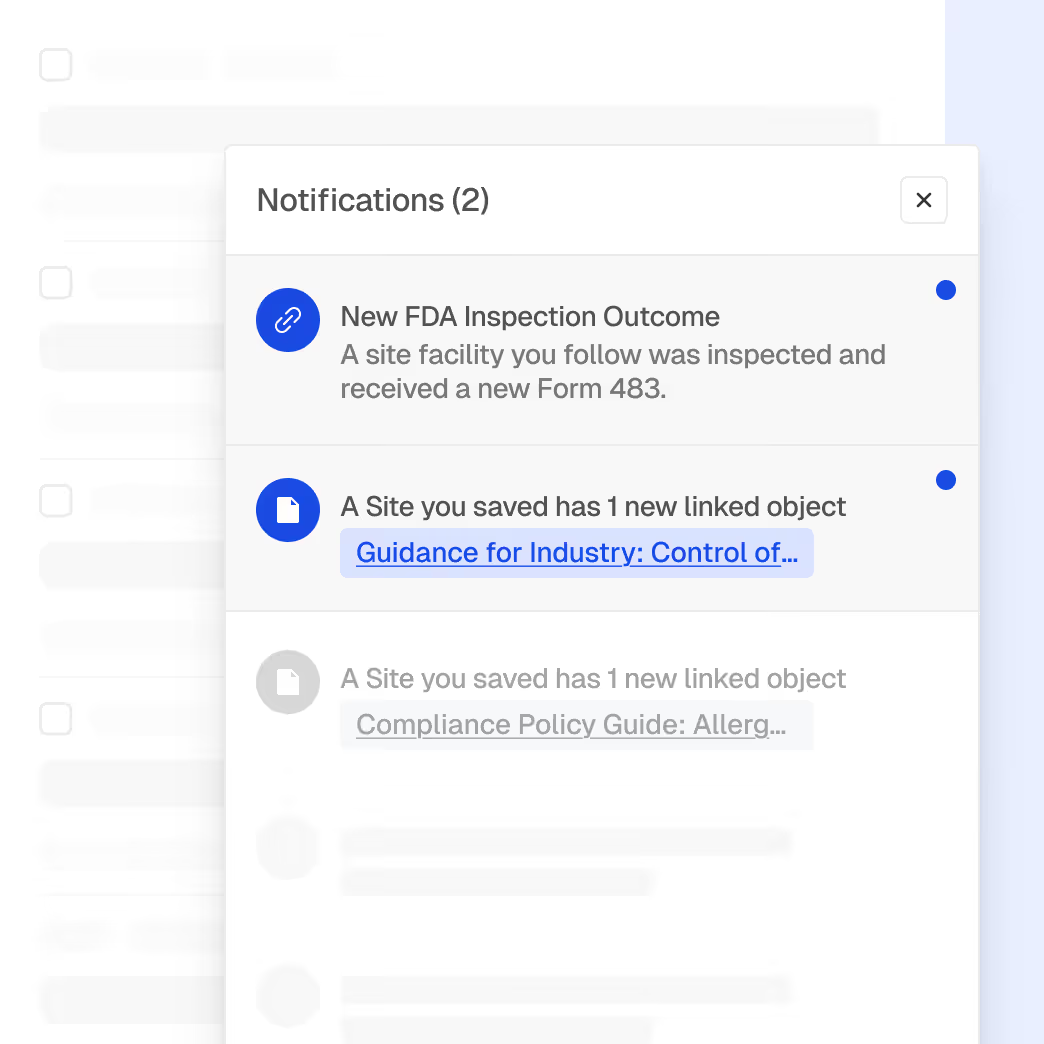
Cross-Portfolio Risk Detection
Identify emerging compliance risks that could impact multiple products or markets.
AI-assisted pattern detection across enforcement actions
Early signals of shifting FDA focus areas
Correlation between regulatory updates and enforcement outcomes
Prioritization support for compliance initiatives
What Makes Redica Different
Ingredient and Claims Risk, Interpreted in Context
Redica tracks how regulators scrutinize cosmetic ingredients, formulations, and marketing claims, helping teams understand where real enforcement risk is emerging.
Early Signals Across Global Markets
AI-enriched intelligence surfaces changes in regional requirements and enforcement behavior early, supporting faster adjustments across global product portfolios.
Visibility Into Supplier and Manufacturing Risk
Redica connects inspection outcomes and enforcement actions to ingredient suppliers and contract manufacturers, reducing blind spots in complex supply networks.
Intelligence That Fits Cosmetics Operations
Access insights through the Redica App or integrate them into QMS, ERP, and supplier management systems to support day-to-day oversight and decision making.
.avif)


