.avif)
Strengthen Global Quality Operations with Connected Intelligence
Quality teams need complete visibility to anticipate inspections, manage suppliers, and identify risks earlier. Redica connects regulatory, inspection, site, and post-market intelligence into a single, explainable source of truth across your enterprise.


















How Quality Teams Can Use Redica
Inspection Readiness
Identify high-risk topics, emerging inspection trends, and regulator focus areas before an audit is scheduled.
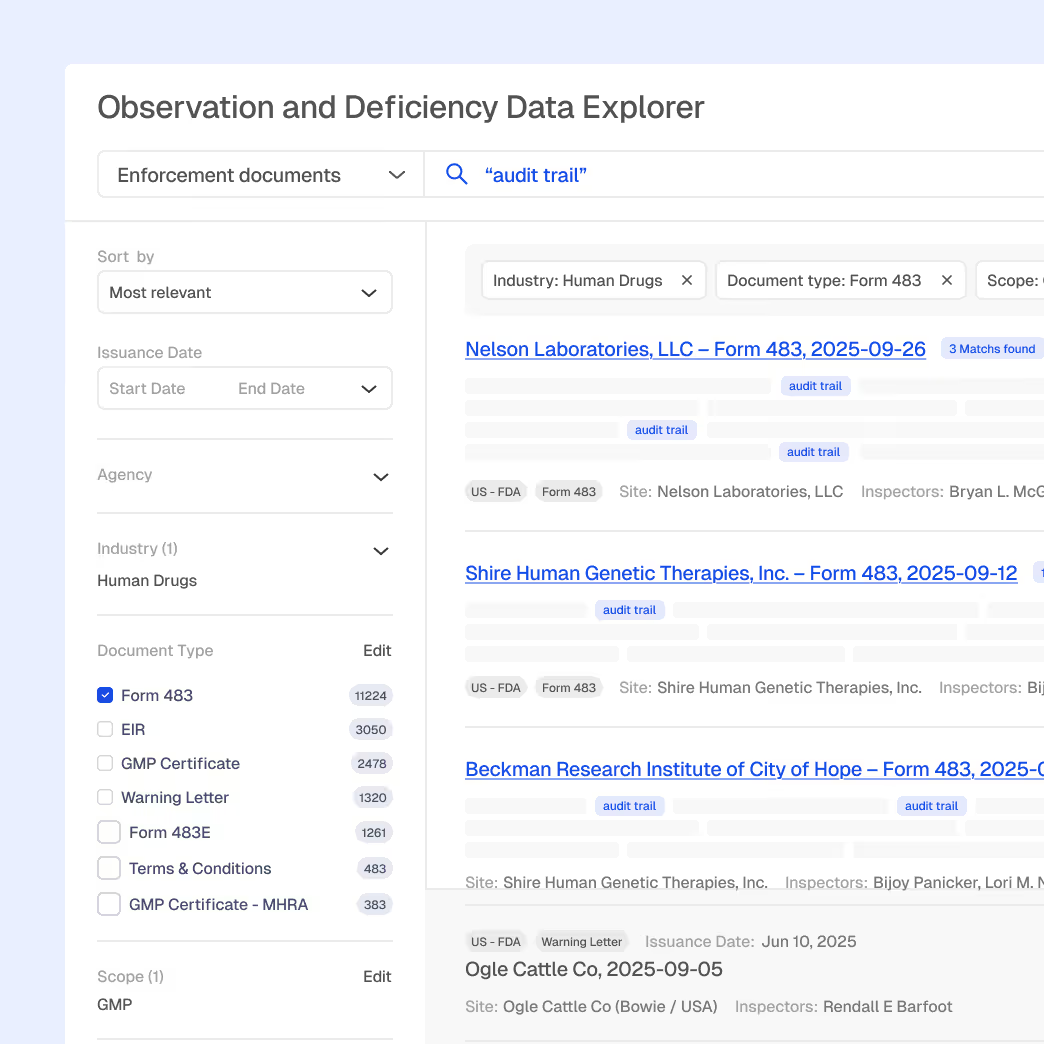
Global inspection and enforcement intelligence
Trending issue analysis by region, agency, and product type
Firm, inspector, and site profiles
Explainable AI summaries for rapid prep
Supplier & CMO Oversight
Monitor suppliers and CMOs more effectively by unifying regulatory actions, inspection history, and site-level risk indicators.
Site Intelligence with risk scoring
Supplier and CMO benchmarking
Alerts for new inspections, actions, or compliance signals
Integrated delivery into QMS/ERP

Monitoring Product Quality & Post-Market Signals
Connect post-market events with inspection findings and regulatory updates to spot emerging issues earlier.
Adverse event, recall, and safety trend monitoring
AI-driven clustering of quality-related signals
Cross-reference against enforcement patterns
Unified timeline view for faster triage
%20(1).avif)
Audit-Ready Change Control
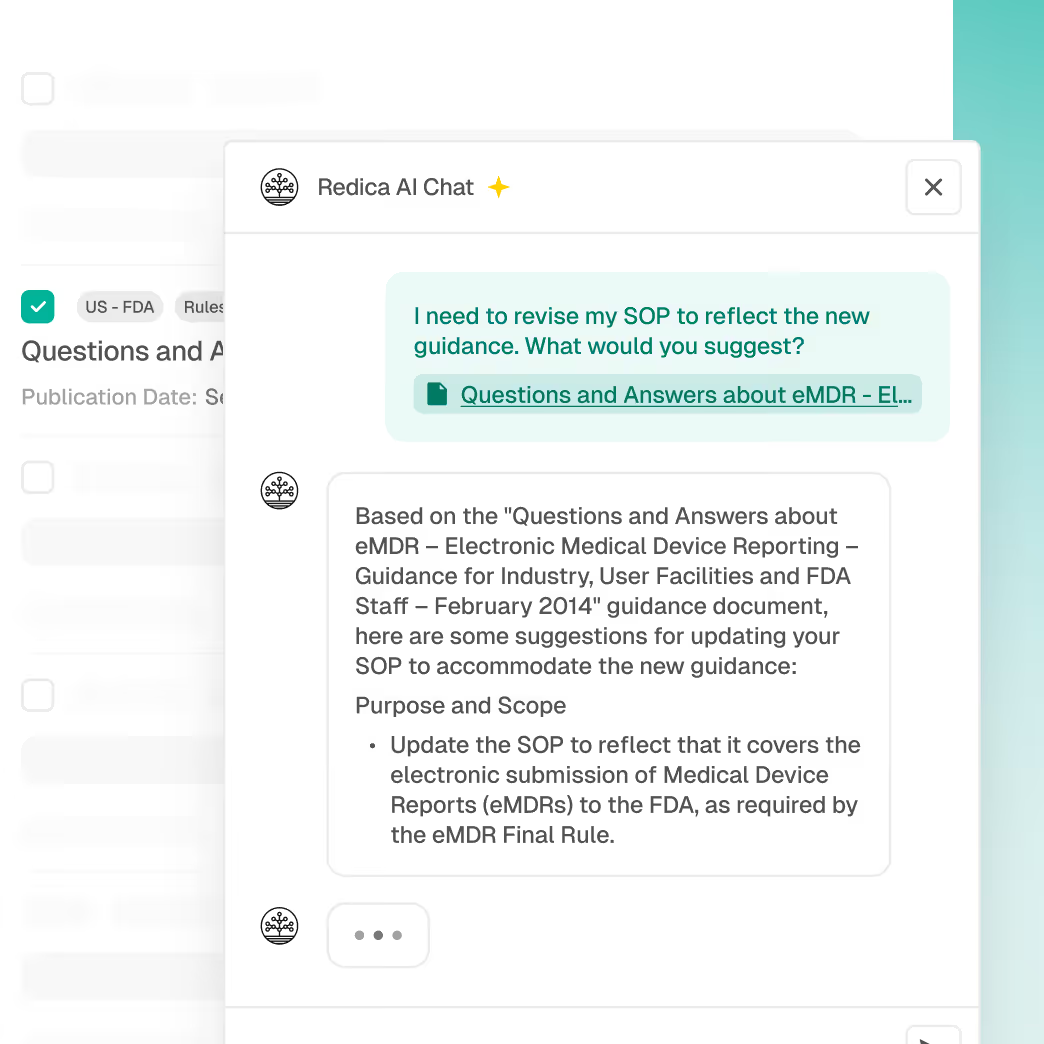
Accelerate change assessments by automatically linking new regulatory updates to relevant SOPs, processes, or product lines.
Regulatory Intelligence with global monitoring
SOP and document impact linking
Redica Copilots for explainable summaries
Traceable citations for audit defense
What Makes Redica Different
A Holistic View of Quality Risk
Redica unifies regulatory, inspection, site, and post-market signals so teams can prioritize risks with full context, not disconnected data.
Explainable, Audit-Ready Intelligence
Every insight includes source citations and reasoning steps, enabling Quality to make faster decisions without compromising compliance.
Designed for Complex, Global Operations
Increase inspection prep efficiency, surface supplier risks faster, and reduce manual regulatory review effort with AI workflows.
Embedded Where Your Teams Work
Access insights through the Redica App or push them directly into QMS, RIM, or ERP systems for seamless operational workflows.
.avif)