
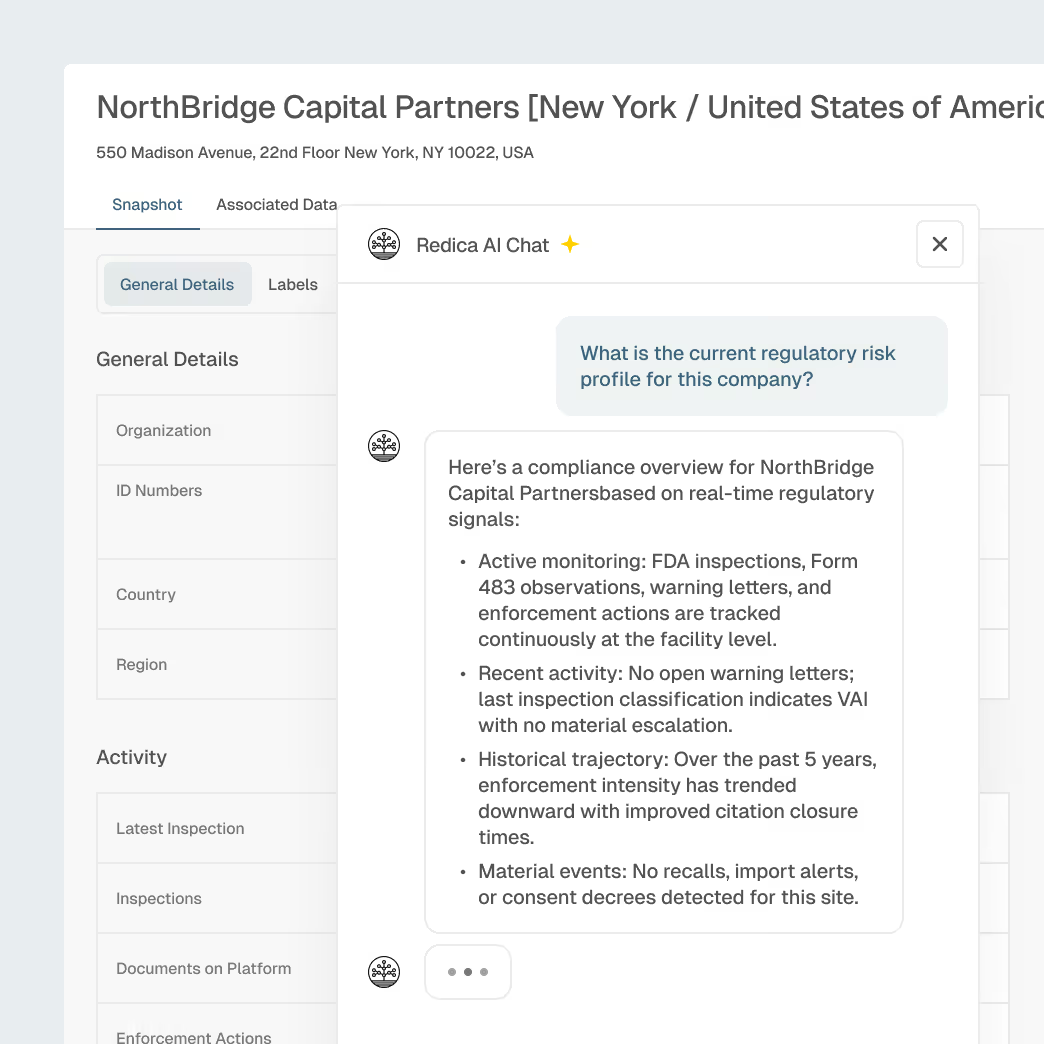
Life Sciences Regulatory Intelligence for Smarter Investment and Risk Decisions
Financial services firms need independent, real-time visibility into regulatory compliance, manufacturing quality, and enforcement trends across pharmaceutical, biotech, medical device, and food companies. Redica delivers explainable regulatory intelligence from FDA inspections, warning letters, recalls, and global enforcement actions, helping investment managers, lenders, insurers, and advisors make faster, more informed decisions.


















How Financial Services Firms Use Redica
Investment Due Diligence and Portfolio Monitoring
Assess regulatory risk and compliance health across life sciences investments.
Real-time monitoring of FDA inspections, warning letters, and enforcement actions
Facility-level compliance intelligence for portfolio companies and targets
Historical enforcement patterns and regulatory trajectories
Material event detection: recalls, import alerts, consent decrees
Explainable AI summaries with source citations for investment memos
Credit Risk Assessment and Lending Decisions
Evaluate borrower quality and identify early warning signals of compliance deterioration.
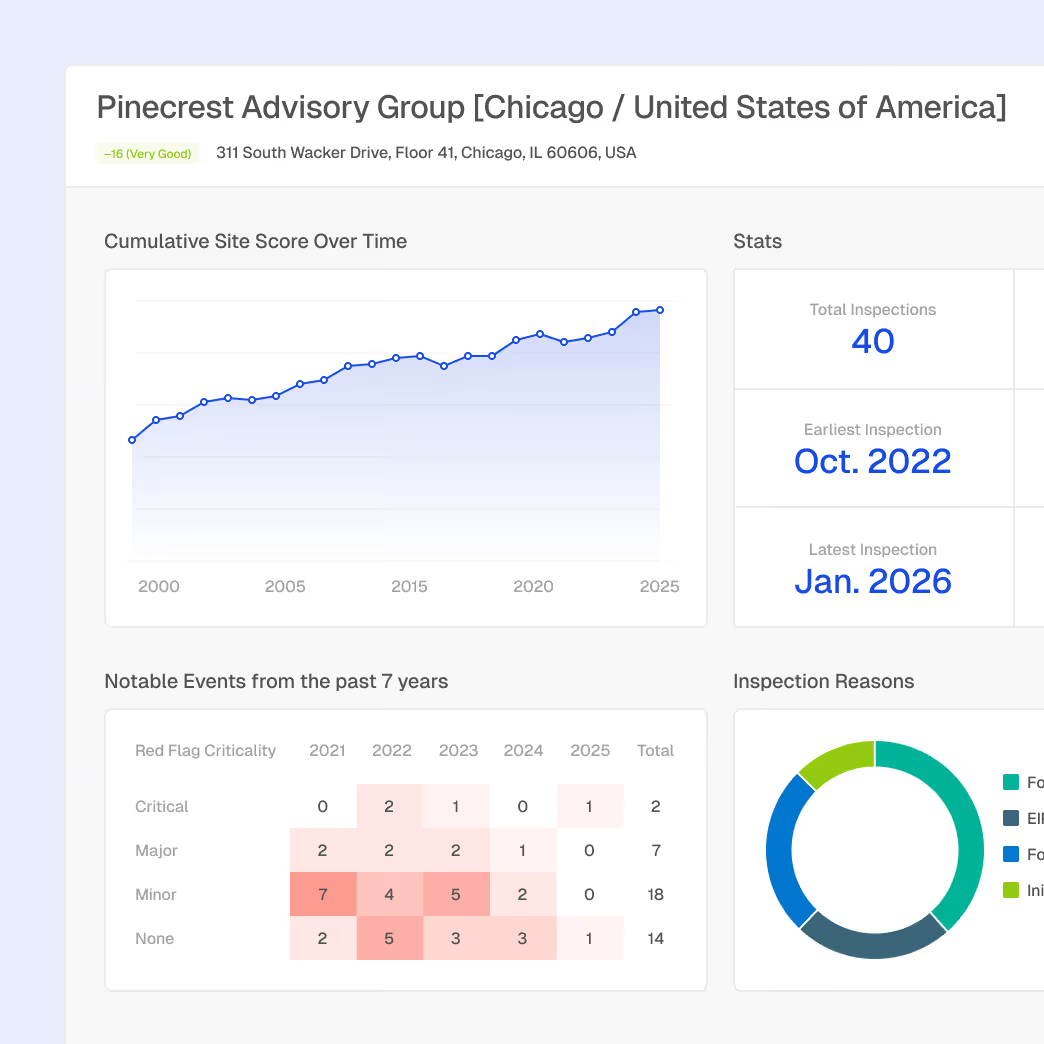
Manufacturing facility inspection histories and quality trends
Enforcement action monitoring tied to borrower entities
Supply chain risk signals from contract manufacturers and suppliers
Correlation between regulatory events and business disruption risk
Objective, data-driven inputs for underwriting and covenant monitoring
M&A and Transaction Support
Accelerate regulatory due diligence and uncover hidden compliance liabilities.
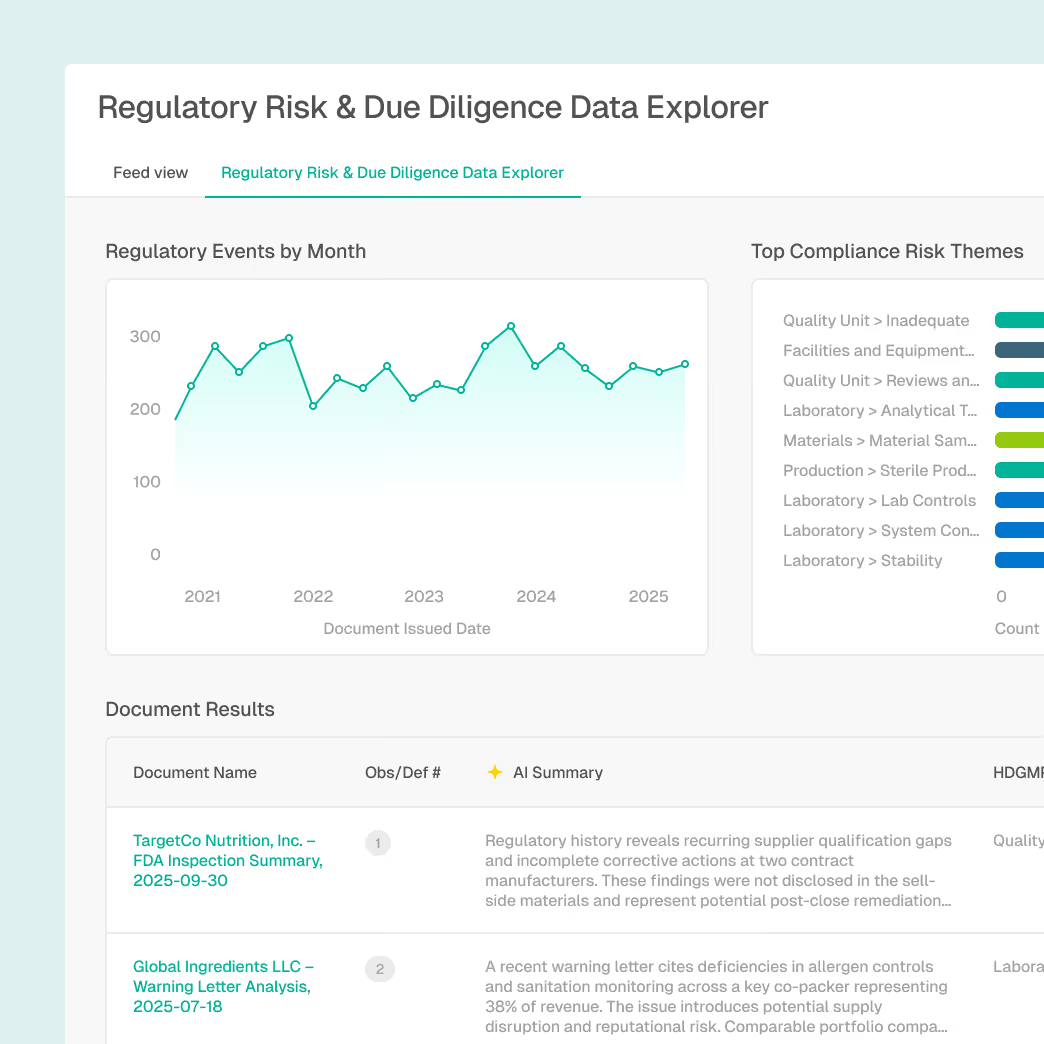
Comprehensive regulatory history for target companies and their facilities
Supplier and contract manufacturer compliance intelligence
Identification of undisclosed regulatory risks and material events
Peer benchmarking for compliance performance assessment
Audit-ready documentation with full source provenance
Insurance Underwriting and Claims Intelligence
Price risk accurately and investigate claims with regulatory evidence.
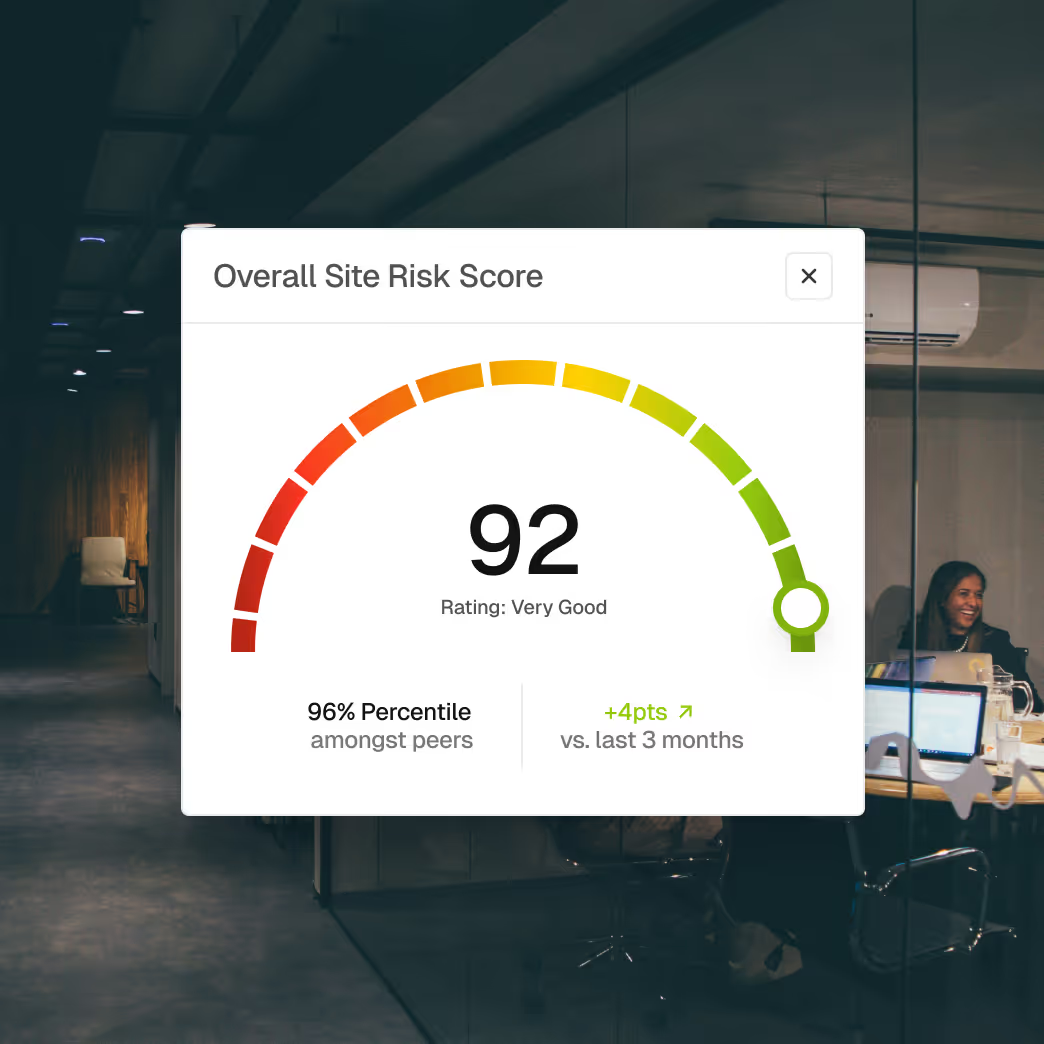
Inspection and enforcement history for underwriting decisions
Compliance pattern analysis for policy pricing and renewal
Claims investigation support with FDA enforcement documentation
Risk segmentation by facility type, product category, and regulatory history
Loss prevention insights from industry-wide enforcement trends
What Makes Redica Different
Independent, Primary Source Intelligence
Redica aggregates data directly from FDA and global health authorities—not company disclosures. Every insight traces to official inspection reports, warning letters, and enforcement actions.
Real-Time Detection of Material Events
AI-powered monitoring surfaces regulatory events as they happen: facility inspections, warning letters, recalls, import alerts, and consent decrees.
Entity-Level Intelligence Across Complex Organizations
Redica ID connects regulatory data across parent companies, subsidiaries, manufacturing sites, and contract partners—revealing risk across entire corporate structures.
Built for Financial Services Workflows
Access through the Redica app or integrate data feeds into investment platforms and risk systems. Every data point includes citations, timestamps, and provenance for audit requirements.
.avif)


