
Turn Post-Market Signals Into Actionable Intelligence
Post-Market Intelligence helps Quality, Compliance, and Safety teams manage vigilance obligations, detect product issues earlier, and connect real-world signals with regulatory, inspection, and enforcement context. Redica AI supports GVP-aligned signal management and faster, more confident decisions across the product lifecycle.
.png)









Unified Responses to Emerging Risks
After launch, product performance and safety signals span adverse event reports, recalls, complaints, field actions, and global vigilance databases. Teams responsible for GVP, quality oversight, and post-market surveillance often struggle to unify these inputs, assess materiality, and understand how signals relate to inspection history or regulatory expectations. Fragmented systems make it difficult to prioritize investigations, support timely escalation, or consistently trigger CAPAs.
Redica AI unifies post-market data from diverse sources, enriches it with inspection and regulatory context, and transforms these raw signals into connected, explainable insights. This empowers teams to detect meaningful trends sooner, align on impact, and coordinate corrective and preventive actions with full traceability.
Post-Market Intelligence to Expose Risk Before It Escalates
Unified Signal Ingestion Across Vigilance Sources
Aggregate adverse events, recalls, field actions, and safety notices from global authorities into a normalized view. Signals are structured by product, site, geography, and issue type to support consistent GVP-aligned review.

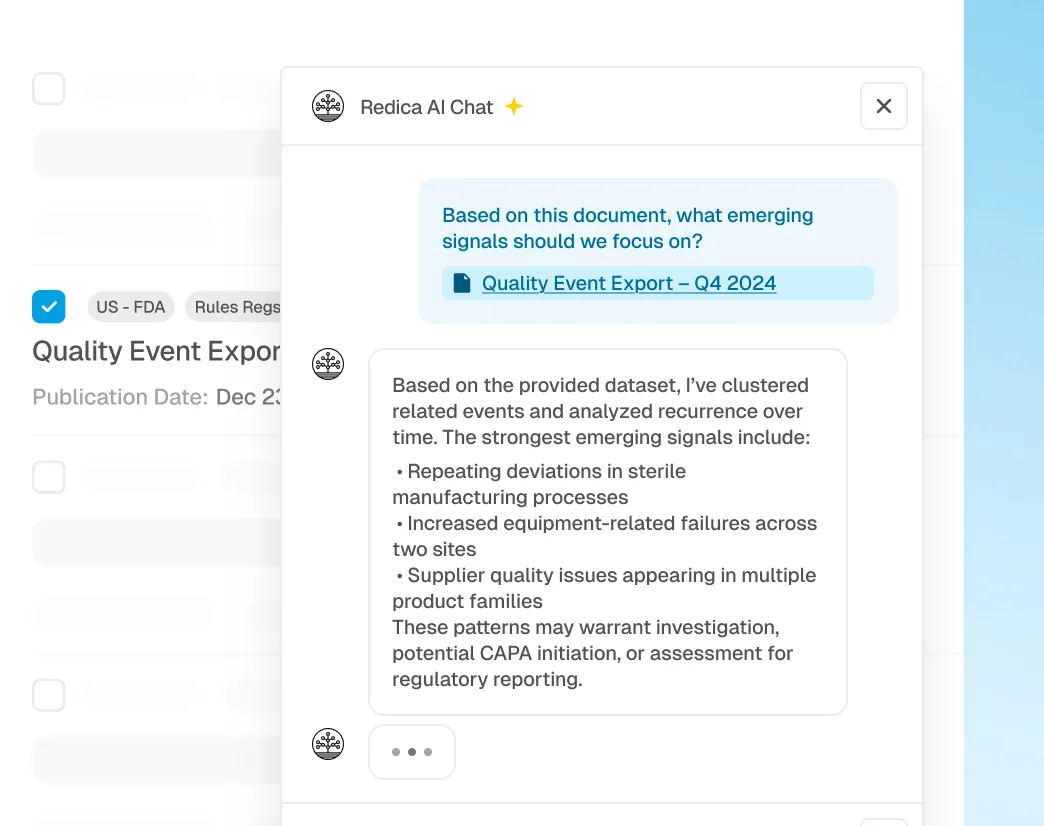
AI-Driven Signal Clustering and Trend Detection
Redica AI groups related events, tracks frequency and recurrence over time, and surfaces emerging signal patterns that may require investigation, escalation, or regulatory reporting.
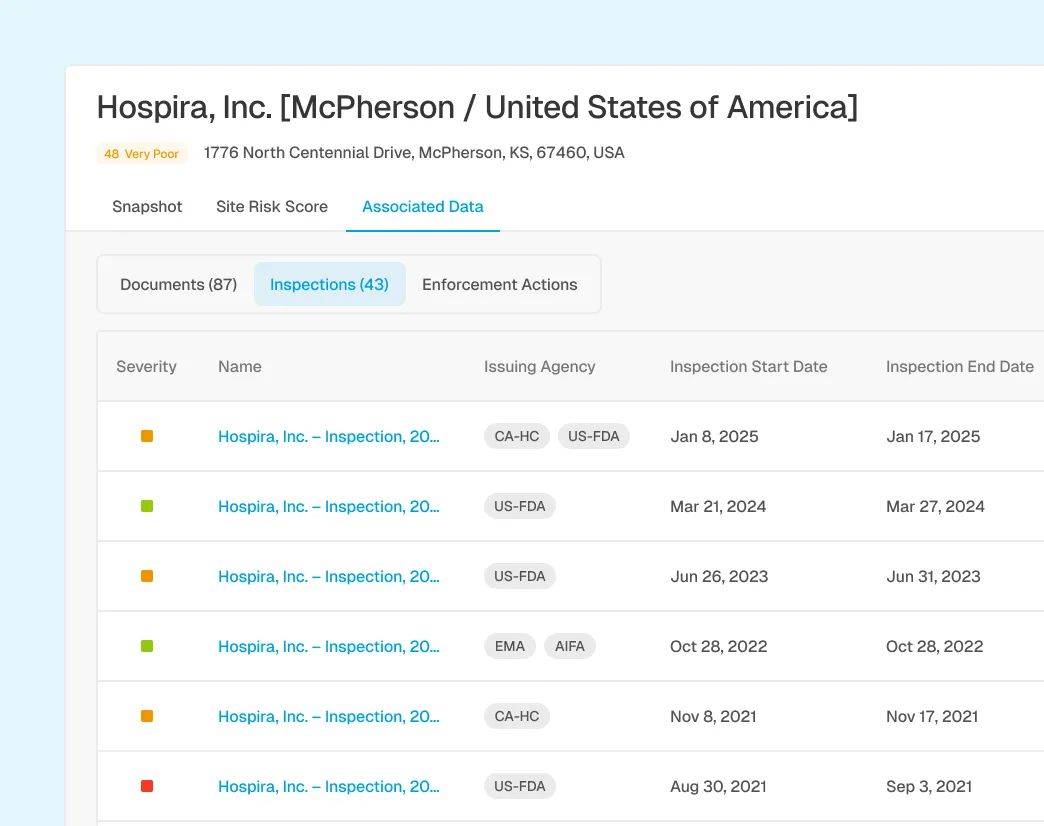
Correlation with Inspection and Enforcement History
Connect post-market signals to inspection findings, warning letters, and enforcement focus areas to identify potential quality system gaps and regulatory exposure.

Contextual Risk Assessment with Explainability
Each signal cluster is enriched with severity, recurrence, spread, and regulatory relevance, with transparent citations and reasoning to support defensible prioritization.
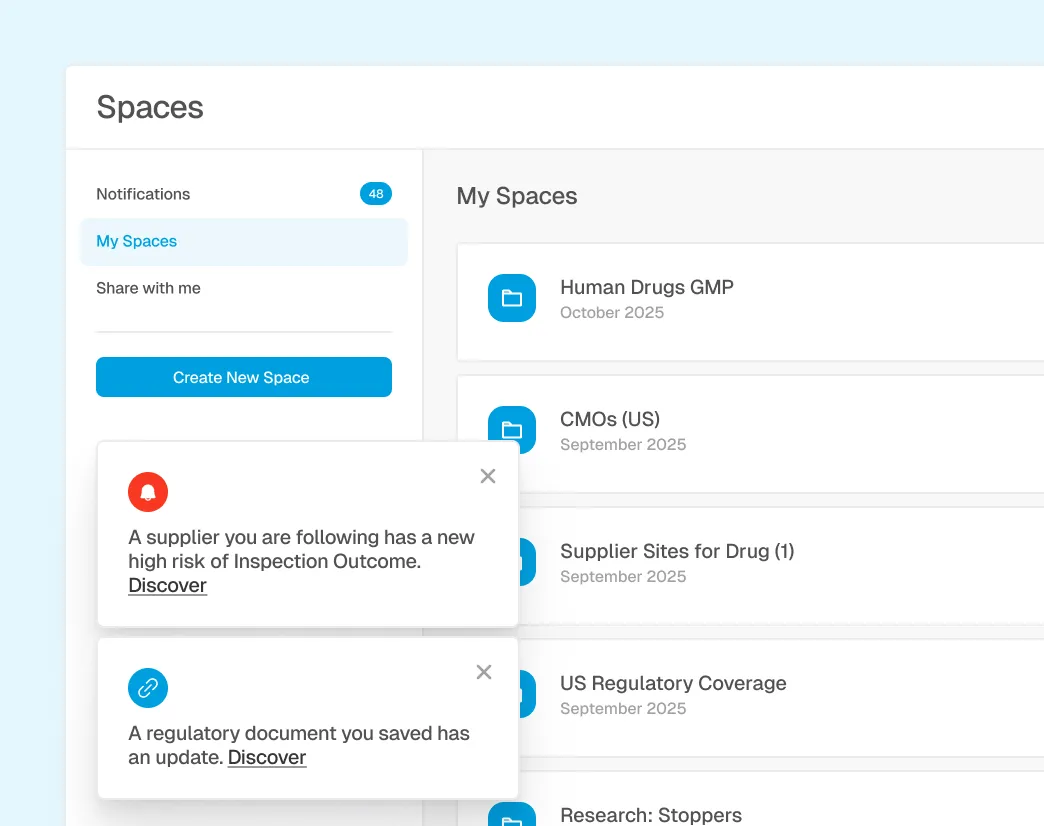
CAPA-Ready Collaboration and Action Workflows
Review signals in shared Spaces, document assessments, assign follow-ups, and link outcomes to CAPA, supplier actions, or regulatory response.
Post-Market Intelligence for Every Function
GVP Signal Management and Surveillance
Identify meaningful safety and quality signals across products and markets to support ongoing vigilance and escalation decisions.
Link Signals to Inspection and Regulatory Risk
Understand how post-market issues align with inspection history, enforcement trends, and regulatory expectations.
Support CAPA & Risk Mitigation Planning
Use post-market insights to inform corrective and preventive actions, link evidence to quality systems, and build more resilient response plans.
This is a powerful analytics tool we use routinely across the full GxP spectrum to monitor supply chain risks, assess vendors, and clinical trial sites. The analytics allow for effective and efficient compliance benchmarking and QMS planning in an easy-to-use format.
Vice President, Global Head of Quality

Discover Post-Market Insights with Redica
Precision rate labeling and reviewing document signals
Connected version history for all new documents
publish of warning letters from FDA posting
Day translation of all non-English documents
Global content ingestion and change detection
Documents summarized with Redica AI
FAQ
Can it integrate with our systems?
Yes. Redica Post-Market Intelligence can be accessed through the Redica App or delivered to QMS, RIM, ERP, and data platforms like Snowflake.
Can Redica be used for CAPA and risk management?
Yes. Teams can use post-market signals to feed CAPA workflows, risk assessments, and corrective planning directly within shared Spaces.
Does it support AI explainability?
Yes. Every AI-assisted insight includes transparent source citations and reasoning steps to support traceable, audit-ready decisions.
How often is the data updated?
Post-market data is updated as new adverse event reports, recalls, and field actions are published by global authorities, with Redica AI agents ingesting and enriching new records continuously.
What makes Redica different from other post-market intelligence tools?
Most systems surface isolated safety or complaint data. Redica unifies post-market signals, enriches them with context from inspections and regulations, and applies AI to highlight emerging patterns that teams can act on earlier.
Still have questions? Contact us



