
Manage Consumer Health Risk with Connected Intelligence
Companies operating in supplements and non-prescription products face evolving regulations, heightened enforcement scrutiny, and complex supplier networks. Redica connects regulatory, inspection, site, and downstream signals into a single, explainable source of truth to help teams detect risk earlier and respond with confidence.


















How Supplements & OTC Teams Use Redica
Regulatory and Claims Monitoring
Stay ahead of regulatory updates, guidance changes, and enforcement trends that impact product formulation, labeling, and claims.
Global monitoring of regulations, guidance, and enforcement actions
Topic-based filtering for ingredients, claims, and product categories
AI-assisted summaries to assess relevance and impact
Traceable citations to support internal review and decision making
Inspection and Enforcement Readiness
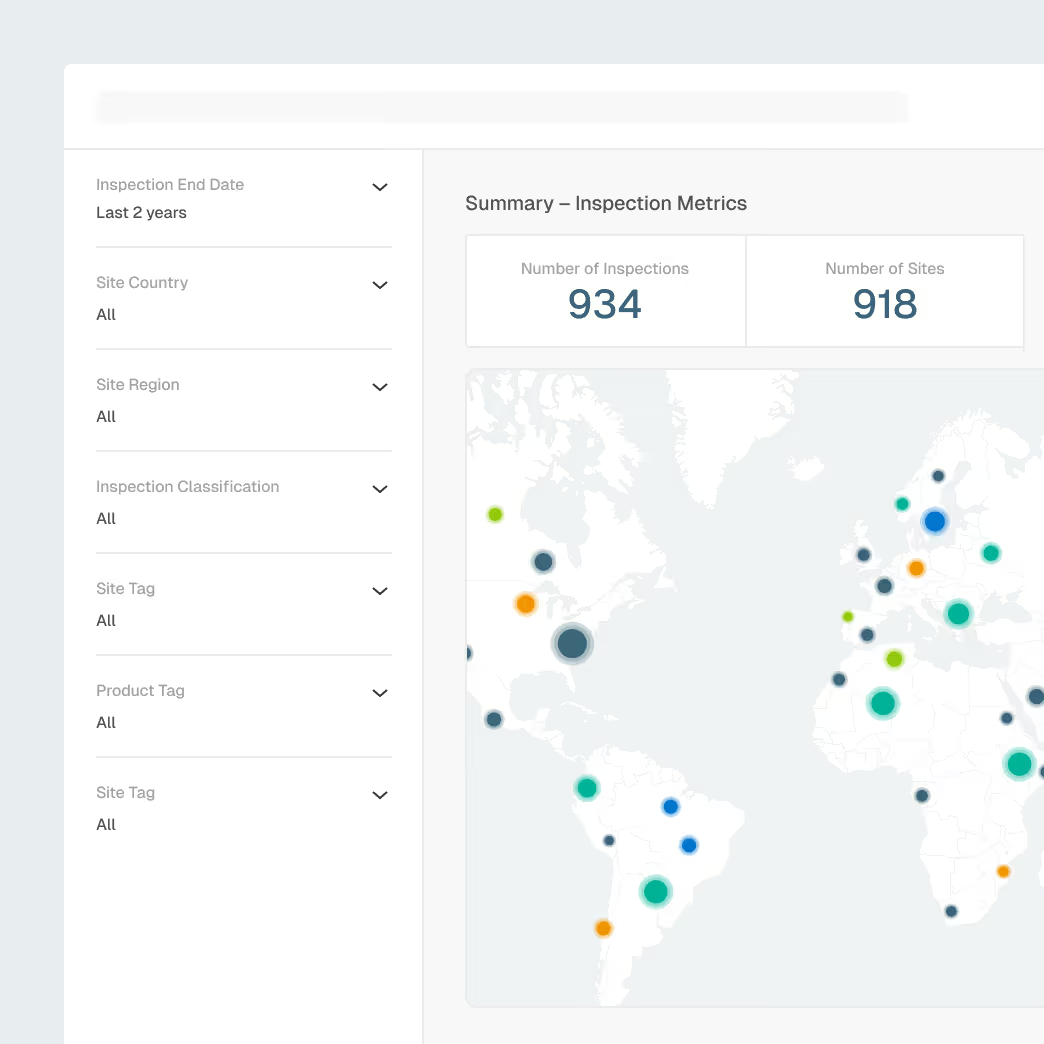
Prepare for inspections and regulatory inquiries with visibility into agency focus areas and historical enforcement behavior.
Global inspection and enforcement intelligence
Site and firm inspection histories
Trending citation analysis by product type and region
Explainable AI summaries for faster preparation
Supplier and Contract Manufacturer Oversight
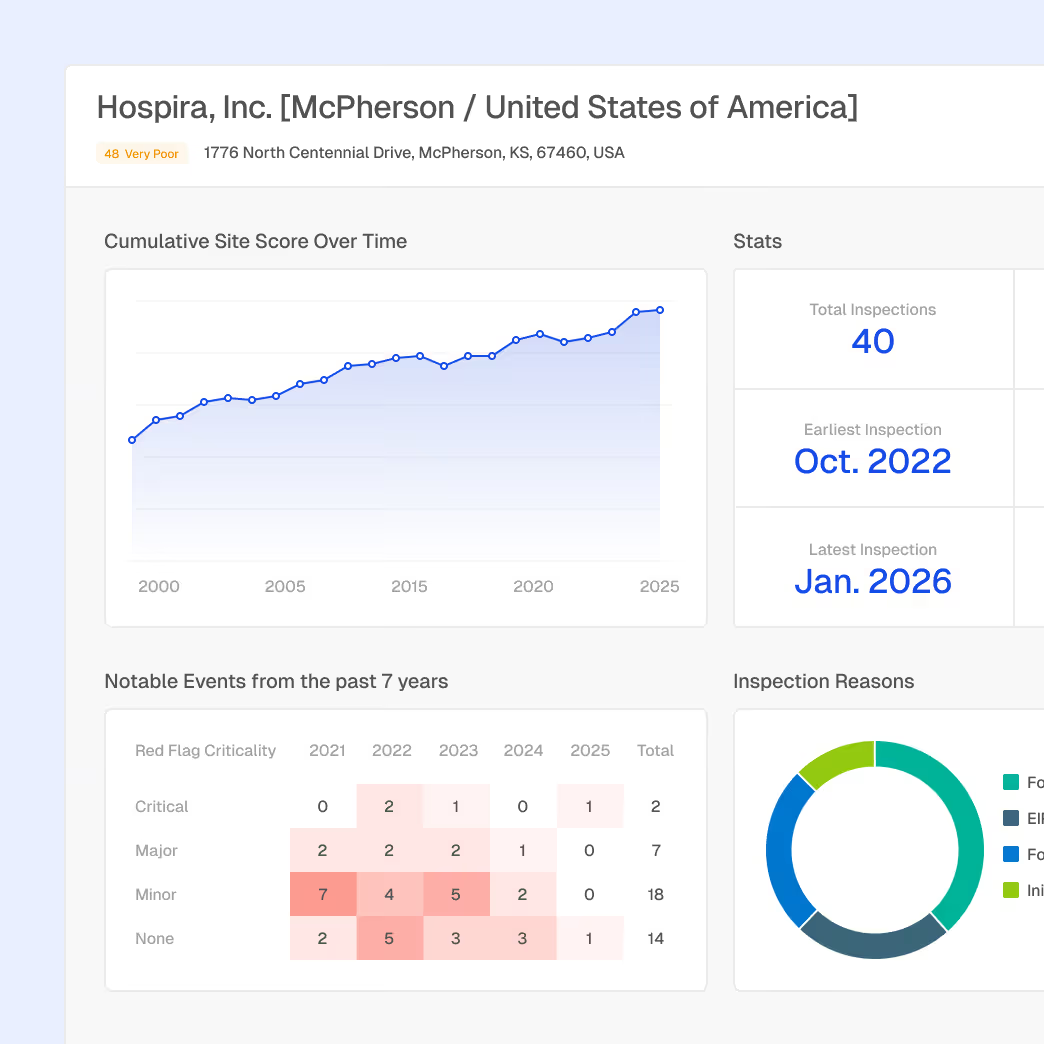
Monitor suppliers and contract manufacturers to reduce quality risk and supply disruptions.
Site Intelligence with supplier and manufactuerer profiles
Inspection and enforcement history tied to facilities
Early signals of emerging compliance risk
Benchmarking to support qualification and ongoing oversight
Downstream Signal and Market Risk Detection
Surface emerging consumer and market risk by connecting recalls, complaints, and marketplace signals with regulatory and enforcement context.
Monitoring of recalls, import alerts, and market withdrawals
Early signals tied to labeling, ingredient, or claims-related enforcement
AI-assisted grouping of recurring consumer and quality complaints
Unified timeline linking market signals to inspection and enforcement history
.png)
Why Supplements & OTC Companies Choose Redica
Enforcement-Led Risk Visibility
Redica helps teams understand how regulators actually enforce requirements across ingredients, claims, and manufacturing practices, not just what guidance says.
Faster Insight for Rapidly Evolving Rules
AI-enriched intelligence highlights relevant regulatory updates and enforcement signals early, supporting quicker internal review and response.
Built for Supplier-Driven Risk
Redica standardizes site and supplier intelligence across contract manufacturers, ingredient suppliers, and private-label partners to reduce hidden risk.
Operational Intelligence That Fits Your Business
Access insights in the Redica app or integrate them into QMS, ERP, and supplier management systems to support day-to-day oversight and decision making.
.avif)


