
Control Outcomes with AI Inspection Intelligence
Inspection Intelligence gives Quality and Compliance teams the clarity to anticipate enforcement risks, recognize regulator focus areas, and take early action. Redica AI enriches global inspection data with context and guidance so teams can strengthen readiness and reduce repeat observations across sites and suppliers.









Proactive Inspection Readiness
Inspection activity is rising across global agencies, yet most teams still rely on static reports, fragmented PDFs, and inconsistent local processes that show only what happened, not what is emerging or why. Without timely visibility into inspector behavior, trending violations, or cross-site performance patterns, organizations struggle to prepare consistently or prevent recurring findings.
Redica AI centralizes and enriches global inspection and enforcement data, applying AI agents and AI-assisted workflows to transform scattered reports into trusted, actionable insights for more predictable and proactive inspection readiness.
Power Consistent, Confident Inspection Outcomes
Global Inspection & Enforcement Data
Access 483s, EIRs, warning letters, inspection narratives, and global agency outcomes in one place. Redica AI agents classify and connect this data so teams can quickly align on where enforcement is increasing and coordinate next steps in shared Spaces.
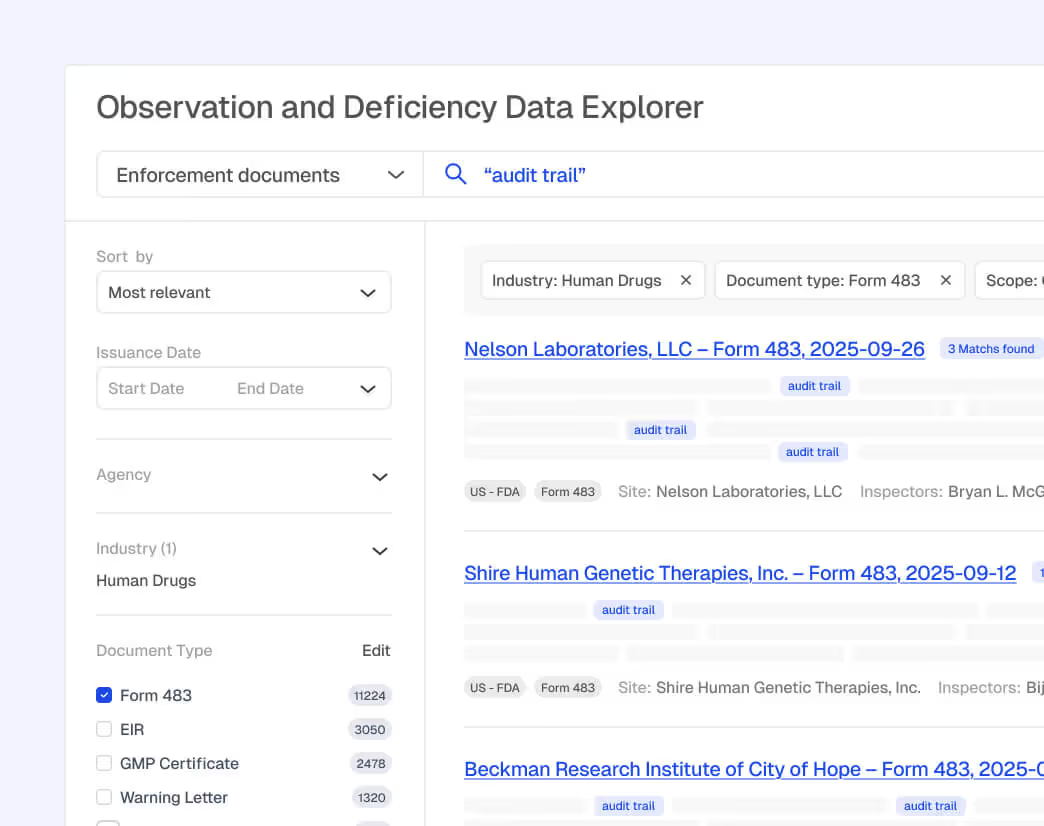

Early Signals for Risk Detection
AI copilots detect emerging violations and regulator-specific trends. Teams can review signals together in Spaces, collaborate on impact assessments, assign owners, and prioritize audits or remediation efforts with shared context.
Context-Aware Evaluation
Redica AI incorporates topic alignment, severity, recency, and relevancy to highlight behavior patterns and enforcement themes. Cross-functional teams can compare interpretations, annotate findings, and make readiness decisions collaboratively.

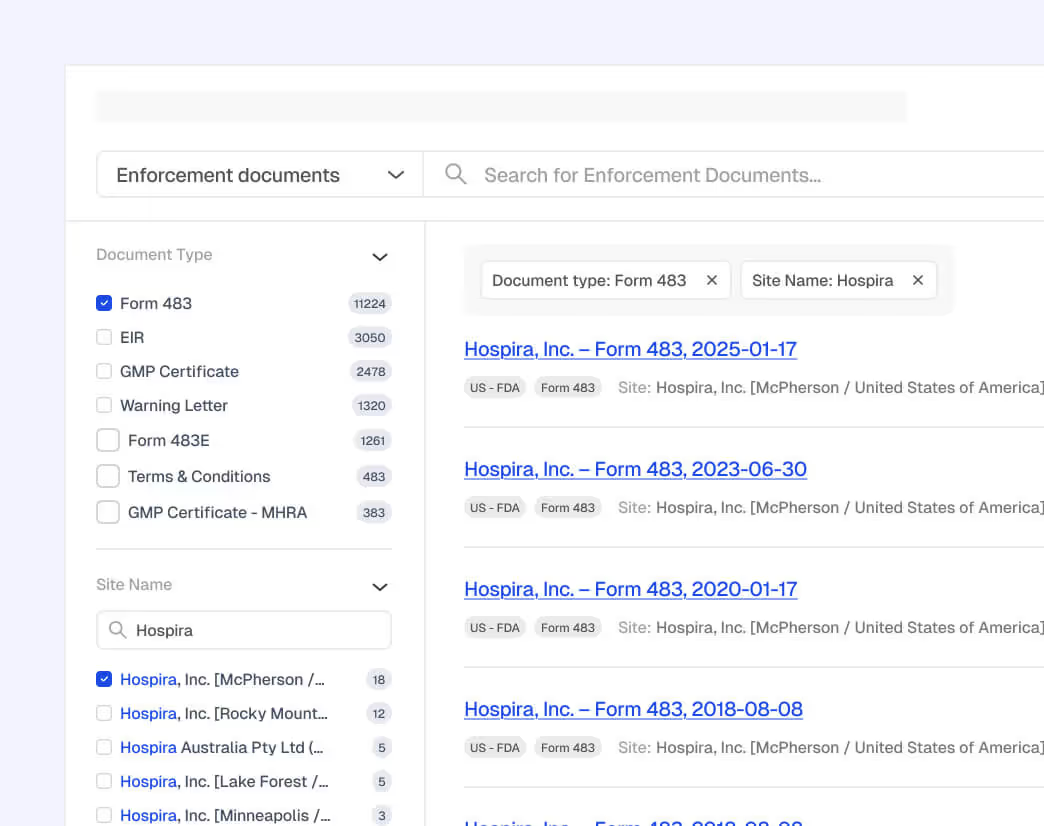
Explainable, Audit-Ready Intelligence
AI-assisted insights include full provenance, citations, and reasoning so Quality, Compliance, and Site teams can validate decisions, document rationale, and maintain defensible, audit-ready records in shared workspaces.
Embedded Intelligence with Veeva and QMS Integrations
Work in the Redica app, integrate intelligence directly into QMS workflows, or leverage Redica integrated solutions that process and connect your internal company data for more streamlined, holistic oversight.
Inspection Intelligence for Every Function
Benchmark and Prioritize Risk
Use AI-enriched inspection trends to benchmark performance, identify vulnerabilities, and focus audit resources where they matter most.
Prepare Sites with Regulator-Specific Insights
Analyze inspector history and enforcement patterns so readiness plans reflect real regulator behaviors, not assumptions.
Strengthen Supplier Oversight
Evaluate suppliers and CMOs using AI-assisted risk indicators built from global inspection histories, enforcement language, and site-level context.
Redica is an essential tool for our quality unit. I know our team will be fully prepared if an investigator arrives at our facility tomorrow. No guesswork here. With a simple search I can have enforcement trends by specific keywords presented quickly and clearly.
Director of Quality, Amgen

Enforcement Intelligence at Scale
Inspection documents including 483s, EIRs, CRLs, and Warning Letters
Global regulatory agencies monitored
Form 483s with observation text and AI summaries
Inspection records across global agencies
Publish of Warning Letters from FDA posting
Refreshed sources annually for proactive, relevant change monitoring
Enforcement documents published monthly
FAQ
Can it integrate with our systems?
Yes. Inspection Intelligence and Redica AI insights can be used in the Redica App or delivered into QMS, RIM, ERP, Snowflake, and document control systems.
Does it support AI explainability?
All AI-driven insights include transparent source citations and reasoning steps for audit-ready traceability.
Does Redica provide enforcement intelligence alongside regulatory updates?
Yes. AI-powered trend detection and inspector analytics highlight where regulators are placing attention so teams can prepare accordingly.
How often is the data updated?
Continuously. Redica AI agents ingest and enrich inspection records, citations, and warning letters from major global agencies.
What makes Redica different from other inspection tools?
Most tools list inspection outcomes. Redica AI connects, classifies, and contextualizes global inspection and enforcement data to reveal emerging risks, regulator behaviors, and actionable patterns across your network.
Still have questions? Contact us



