Table of contents
Are you certain that your cleaning validation processes would pass muster during your next inspection?
Inspectors expect to see that your GMP operations include robust cleaning validation. This ensures that cleaning processes do not result in residue that could potentially contaminate the product.
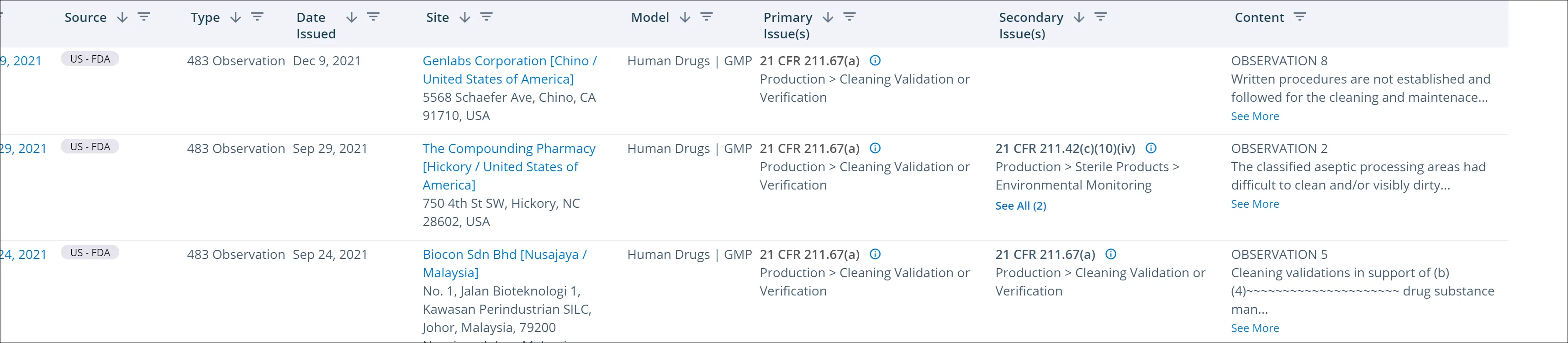
What happens if your cleaning validation processes are found to be inadequate? Cleaning validation and other issues related to cleaning are often cited in FDA warning letters. For example, an FDA investigator who has conducted human drug GMP inspections has found fluid bed dry cartridge filters with powder residue in spite of being labeled as clean, powder residue on several pieces of manufacturing equipment, and “clean” equipment with water dripping off it.
This same investigator also referred to another inspection involving an encapsulator at a global manufacturer of tablets and capsules. Here, cleaning processes on a multiproduct line failed to prevent a yellow powder from potentially contaminating another product consisting of a white powder.
Cleaning Validation: Also an Issue for Medical Devices
In addition to human drug GMP operations, medical device manufacturers are also advised to stay on top of cleaning validation.
Cleaning issues seen during inspections of medical device manufacturing facilities include:
- Failure to identify potential contaminants that could be introduced during manufacturing
- “Visually clean” requirements that involve implantable devices
- Lack of documentation for worst-case device requirements
- Inadequate process characterization
- Test method validation issues
Whether you are a manufacturer of human drugs/biologics or medical devices, regulators expect you to have strong cleaning validation practices that prevent contamination and cross-contamination. Otherwise, your site risks receiving an enforcement action such as a 483 or warning letter.
Did you know that Redica Systems can provide you with access to 483 observations that include cleaning validation findings? Contact us today to learn how you can have access to the largest database of 483 observations.
Additional Cleaning Validation Resources
Inadequate Equipment Cleaning Leads to Cross Contamination, FDA Warnings


