Table of contents
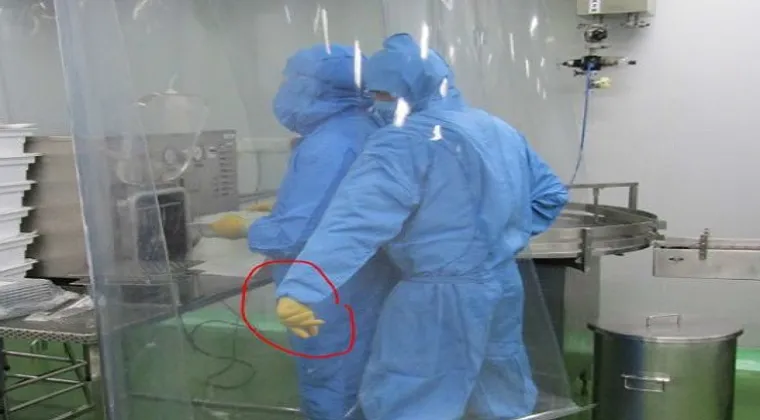
At the International GMP Conference held virtually in March 2022 and co-sponsored by the University of Georgia at Athens and FDA, FDA Office of Regulatory Affairs National Drug Expert Captain, Ileana Barreto-Pettit, presented in-depth case studies from recent drug GMP inspections that illustrate agency findings and concerns.
In a four-part series of articles, Redica Systems Senior GMP Quality Expert Jerry Chapman dove into each case study presented, including an in-depth analysis of the findings, lessons learned, and how companies can avoid similar shortcomings.
Areas examined in the case studies are:
- Inadequate change management systems
- Cross-contamination in solid oral dosage facilities
- Inadequacies and potential contamination hazards in aseptic manufacturing
- Perforated tables and work surfaces in cleanrooms
Download all 4 Case Studies in a Compiled Report
Easily read and share all four case studies by downloading the compiled report by Jerry Chapman. Click here for this FREE instant download.