

Report – A Deeper Dive into FDA’s Newly Published Complete Response Letters (CRLs)
A Data-Driven Look at FDA Complete Response Letters: Trends, Risks, and Insights
Using Redica’s advanced analytics, the report highlights the key risk factors shaping CRL outcomes, from manufacturing readiness and GMP performance to inspection history and site oversight. Learn why more than half of all NDAs and BLAs receive a CRL and how early visibility into inspection risk can transform your regulatory strategy.
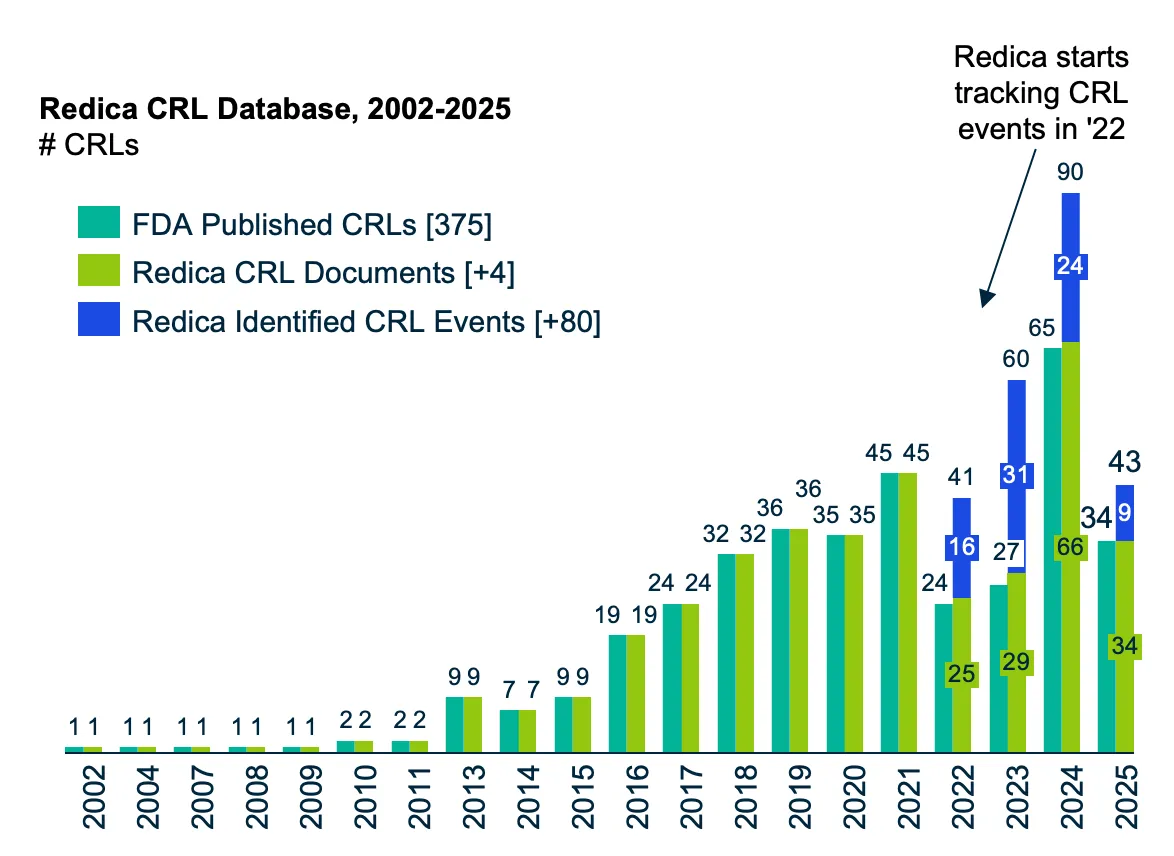
Download your copy of the report to explore FDA’s new era of transparency, understand the patterns driving CRL issuance, and gain the intelligence needed for smarter, faster regulatory success.









If you have trouble with the form, please contact events@redica.com.