November 30, 2020

category
Russian Drug Regulator, MHRA, and FDA Processes for Evaluating & Escalating Drug GMP Inspection Findings
category
CBER Director Wilson Bryan On How To Cut CGT Product Approval Times In Half
November 18, 2020
.webp)
category
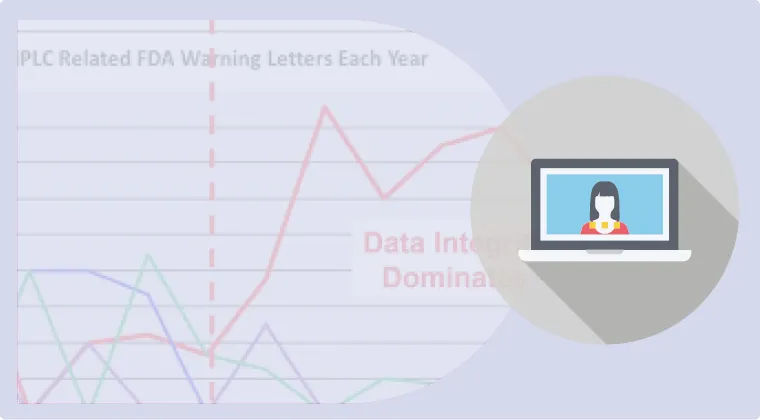
Top 10 Pharma Inspection Findings from FDA, MHRA, and the Russian Drug Regulator
November 11, 2020