Supplier Network Access Guidelines
This Supplier Network Access Guidelines (these “Guidelines”) supplement and are made a part of the Platform Access Agreement (the “Agreement”) and the Supplier Data Network Addendum (the “Addendum”) between Redica (“Redica”, “Us”, or “We”), and an individual or entity (“You”) who accesses or uses Redica’s Services. Capitalized terms used but not defined in these Guidelines shall have the meaning ascribed to them in the Agreement and the Addendum. These Guidelines apply to You, and You agree to be bound by the terms and conditions of these Guidelines in addition to the terms of the Agreement and the Addendum, if You access or use the portion of the Data Network Services. To the extent there is a conflict between these Guidelines and the Agreement or the Addendum, these Guidelines shall not govern.
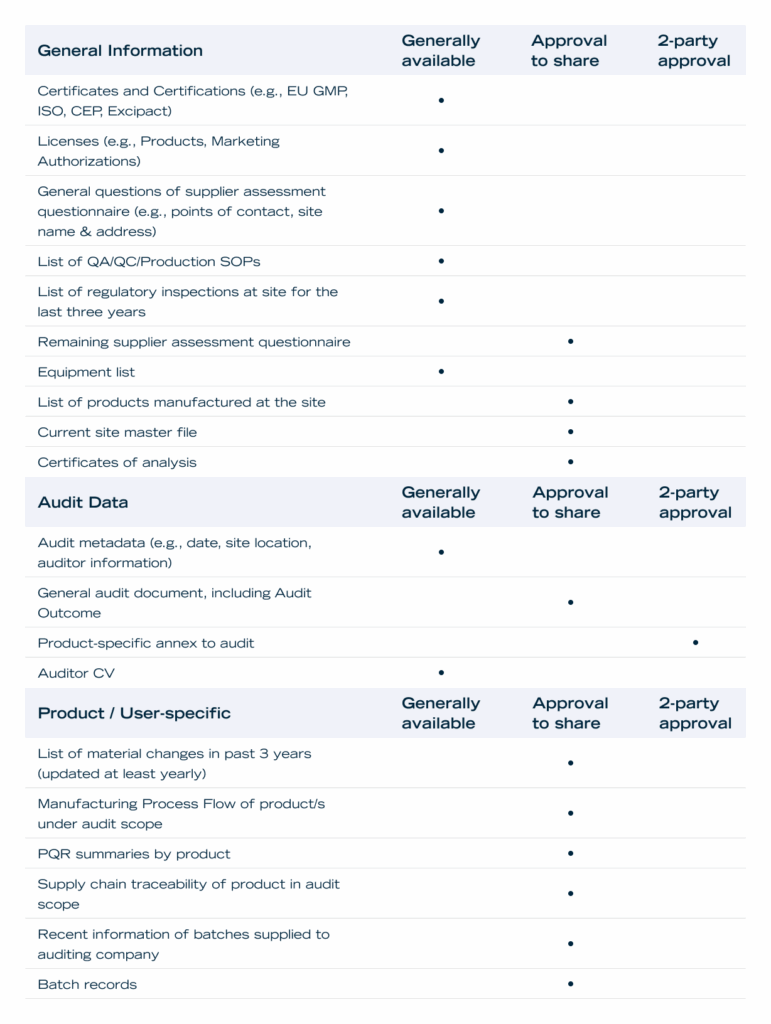
These Guidelines apply to Content and Supplier Documents (as defined in the Addendum, hereinafter collectively referred to as “Content”) You provide as part of an audit as as a Supplier in the Redica Supplier Data Network, and detail which parties will have access to Content uploaded by You. Publicly available information or information obtained through generally public sources is not subject to these Guidelines.
Definitions
Generally available: By uploading Content marked below as “Generally available”, You consent to making the Content available to all other users who have access to Redica.
Approval to share: By uploading Content marked below as “Approval to share”, You consent to making existence of the Content available to all other users who have access to Redica, provided, however, that the Content itself will not be made available to any other users of Redica unless and until You provide affirmative approval. Redica will seek Your affirmative approval on a per user organization / per Content basis. In other words, Your grant of approval for a given Content to a user organization does not grant access to the same Content to other user organizations, nor does such approval grant access to other Content within the same user organization. Once you grant approval to specific Content, such an approval is perpetual.
2-party approval: Content marked below as “2-party approval” is subject to the same terms as Content marked Approval to share, except that it also requires the approval of two parties. Such Content is limited to product-specific audit annexes, which require the affirmative approval of both the audited supplier and the audit initiating company before sharing with any users which were not party to the audit.
Default marked with ‘•’