
Assessment – MAUDE Product Snapshot
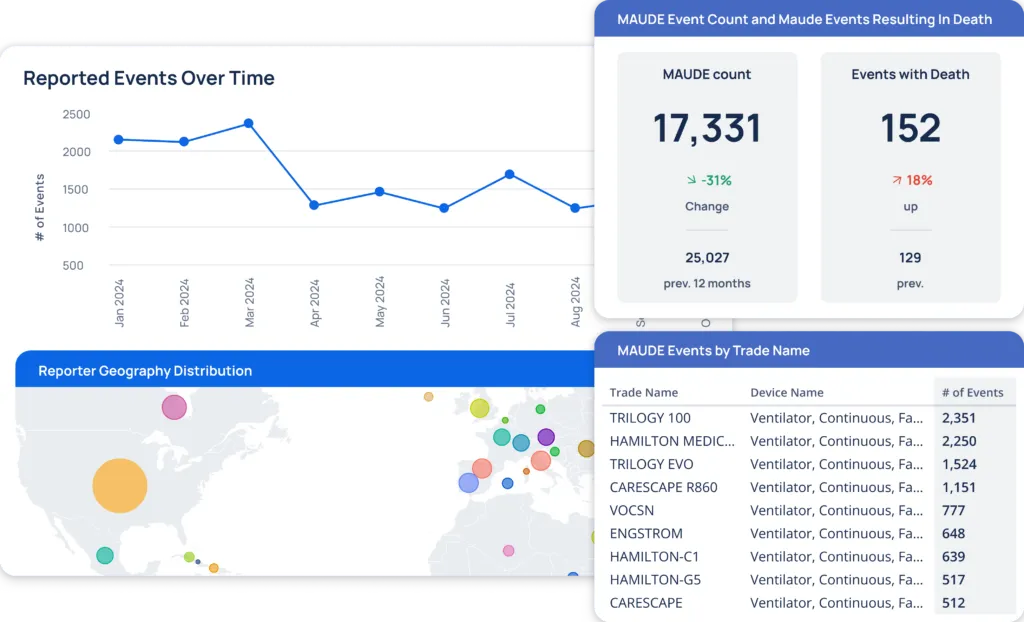
The MAUDE Product Dashboard allows you to view various MAUDE events, including reported incidents over time, sorted by trade name, geography, device issues, and more.
Discover what Redica Systems can offer your medical device company by requesting a MAUDE Product Snapshot for a specific product over the past year. The report will include:
- Total events
- Event trends
- Comparison to similar products
- Product risk profile
Fill out the form, and a team member will contact you to schedule a convenient time to review the data.









If you have trouble with the form, please contact events@redica.com.